Biography
Interests
Mutinta Matambo* & Ravi Paul
Department of Psychiatry, University of Zambia, School of Medicine, Lusaka, Zambia
*Correspondence to: Dr. Mutinta Matambo, Department of Psychiatry, University of Zambia, School of Medicine, Lusaka, Zambia.
Copyright © 2020 Dr. Mutinta Matambo, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
This study explored the verbal fluency performance in adults presenting with epilepsy at University Teaching Hospital. The study was guided by three objectives: (i) to describe verbal fluency performance in adults with epilepsy by the use of the neuropsychological tests; namely Controlled Oral Word Association Test (COWAT) and Category Fluency Test (FAS), (ii) to explore the association between verbal fluency scores and the clinical characteristics of Epilepsy; that is the age of onset and duration of the illness, and (ii) to describe the association between verbal fluency scores and the demographic data (sex and years of education) of the individual with Epilepsy.
The study was quantitative in nature and employed a cross-sectional survey design. Data was collected from participants aged 18 and above at University Teaching Hospital’s (UTH), Clinic 4. The sample size was 32 participants. Systematic sampling was used to select participants into the study. Data was collected through psychological tests: Category Fluency Test (FAS) and Controlled Oral Association Test (COWAT). Data was analyzed using SPSS version 26 and the relationship between variables were tested by use of bivariate analysis.
The study established that patients with epilepsy did not perform well on verbal fluency scores as compared to their counterparts, the respondents without epilepsy. Patients with epilepsy had problems on performance on both the COWAT and Category Fluency Test; with challenges giving the correct responses and with intrusive, perseveration and variant errors being present in their responses. The results further found that, age of onset of the Epileptic illness was seen to impact verbal fluency scores. The study has further established that most of the correlations between verbal fluency scores and years of study, sex of respondent and duration of sickness were found to be insignificant. It was therefore concluded that years of study, duration of sickness and sex of respondents for both epileptic and none epileptic groups did not influence performance on both COWAT and Category Fluency Test.
Background of the Study
For humans, verbal fluency facilitates communication which is a requisite part of everyday life and which is essential in building relationships; in the formulation of one’s self or identity; in play; gaining and sharing knowledge and information and connecting them with all other such important aspects of everyday life, (Harcourt and Brace, 1921). This means that to communicate, an individual will need to be able to effectively and efficiently search for and retrieve words that fit a particular criterion in a reasonable amount of time.
According to Kreutzer et al (2011) [1], verbal fluency is a cognitive function that facilitates the retrieval of various types of information from memory. Kreutzer further states that the ability to correctly retrieve specific information in a given time period calls for control over executive and higher order mental processes such as self-monitoring, internal response generation and selective attention among others. Functional Magnetic Resonance Imaging (fMRI) has shown that these cognitive processes occur as a result of neural mechanisms present in the brain [2]. Therefore it can be surmised that verbal fluency ability has a tangible link to the brain.
Verbal fluency is measured by verbal fluency tests in which participants have to produce as many words as possible in a particular class, in a given amount of time. Due to the fact that verbal fluency refers to the capacity of a person to self-generate, in rapid fashion, phonemic categories (e.g. names of different words beginning with a given letter of the alphabet) or within some semantic category (e.g., animals, fruits, vegetables) [3]; the classes in verbal fluency tests need to be concerned with both phonemes and semantics. Semantic and phonetic fluency are measures of non-motor processing speed, language production, and executive functions in which participants are asked to either generate words in a specific category (e.g. fruits, animals) or words beginning with a specific letter, (Greenaway et al, 2012). Verbal fluency tests have been validated as tests of executive and expressive language ability due to the fact that participants are expected to access their mental lexicon, focus exclusively on the task at hand, select words that meet certain constraints and to avoid repetition [4].
Verbal fluency tests are used in neuropsychology practice to support diagnosis of neuropsychiatric conditions such as Attention Hyperactive Disorder (ADHD) and Dementia or to determine the level and type of cognitive deficits present in certain conditions. One such condition is Epilepsy. Approximately 50 million people currently live with epilepsy worldwide. In rural Kenya, prevalence rates stand at 41 per 1000 (Mungala- Odera et al, 2018); while; in Zambia, 14.5 per 1000 was seen in a catchment area of about 55,000, (Birbeck et al, 2004). The neurology of Epilepsy includes; its causes, the age at which the condition starts, the types of seizures, duration of the illness, etiology, and its treatment. Delving into this aspect is imperative as cognitive problems in Epilepsy can be initiated or exacerbated by causes of the disorders (brain lesions being at the top of the list); seizure type and duration; epileptic dysfunction and treatment [5]. This paper therefore intended to use verbal fluency tests to determine if adults with Epilepsy suffer impairments in verbal fluency; whether the sex of an individual or the clinical factors of the condition impact their verbal fluency levels in any way.
The effects of Epilepsy are multi-faceted and extensive, going beyond seizures and often resulting in
cognitive impairments and social discrimination, (Kerr, 2012). The understanding of, and treatment of these
co-morbidly occurring impairments, though, remains low, mainly due to a lack of knowledge. A lack of
treatment of these impairments affects an individual’s life as the ability to communicate, socialize, and earn
a living is hindered, (Veneers, 2008). A study in North-Western Zambia has shown that epilepsy causes
negative effects on adults. The study looked at cognitive impairment and quality of life of people with
epilepsy and neurocyticercosis. ((Nau et al, 2017). The research found that people with epilepsy faced a
number of cognitive depreciations, (Nau et al, 2017). There exists, though, a gap in knowledge in the study on
the specific cognitive impairment of verbal fluency in adults with Epilepsy in Zambia. It is unclear whether
adults with this condition in Zambia experience declines in verbal fluency. Therefore this study looked into
finding out if there is a connection between Epilepsy and Verbal fluency in adults at the University Teaching
Hospital (UTH).
This study was guided by the following research objectives:
1. To describe verbal fluency performance in adults with epilepsy by the use of the neuropsychological tests;
namely Controlled Oral Word Association Test (COWAT) and Category Fluency Test (FAS).
2. To explore the association between verbal fluency scores and the clinical characteristics of Epilepsy; that
is the age of onset and duration of the illness.
3. To describe the association between verbal fluency scores and the demographic data (sex and years of
education) of the individual with Epilepsy.
Literature Review
A study by Miller et al (2016) studied 38 adults with Epilepsy against 29 healthy controls. In this comparative
study done in the USA at Brown University; both the healthy controls and the adult patients with Epilepsy
completed a neuropsychological battery where it was found that patients with epilepsy performed worse across nearly all cognitive domains, and were clinically impaired (i.e., ≥ 1.5 SD below mean) on more
individual tests when compared with controls, including a subset of patients with epilepsy with normal
MRIs. In addition, Metternich, Buschmann and Schulze-Bonhage (2014) [6] found disfluencies in adults
with Epilepsy. However, the main aim of the paper was to compare SVF and PVF and it also only considered
one kind of epilepsy (focal epilepsy).
A study by Thompson and Duncan (2005) [7] which assessed 136 patients found that Epilepsy did have
an impact of cognitive functioning; including that of semantic verbal fluency and phonemic verbal fluency.
While the research did slightly tackle verbal fluency; it should be noted that the researches’ main focus was
on overall cognitive decline.
Prolonged duration of Epilepsy may also cause a deterioration of general intellectual functioning, (Elger, 2004). According to Iterson et al (2014), the research which used the Wechsler Intelligence Scale to test verbal performance and full scale IQ found that there was a downward progression of function from the first test during earlier periods of Epilepsy to the second test. This shows clearly that the longer a child has the condition, the larger the likelihood of there being intellectual dysfunction and concurrently language deficits.
The age at which an individual begins to have seizures characterized as being epileptic can be related to
cognitive functioning. GDFC (2004) states that the early life of a human being is a period where there
are rapid changes in the brain. During this time the brain forms and refines the complex networks of
connections in the brain through synaptogenesis, pruning, and myelination. It can therefore be justified that
epileptic seizures that begin earlier in life, in childhood, will affect cognitive processes at a higher level than
those which begin later.
A study in Philadelphia in the United States of America (USA) looked at how the early and late age of seizure onset have a differential impact on brain resting-state organization in temporal lobe epilepsy [8]. This study investigated 50 individuals with TLE and 50 aged matched health controls and found that those with a lower age of onset and longer duration caused different perturbations of network modularity and connectivity at the global and local level, with different implications for regional plasticity and adaptive organization. From this it can be deciphered that age of onset and duration of illness do indeed affect brain mechanisms and can consequently influence verbal fluency.
Prolonged duration of Epilepsy may also cause a deterioration of general intellectual functioning, (Elger, 2004). This was proven by a research done by the SEIN Epilepsy Institute, Netherlands, in which 113 children with epilepsy were assessed twice in tertiary epilepsy settings. According to Iterson et al (2014), the research which used the Wechsler Intelligence Scale to test verbal performance and full scale IQ found that there was a downward progression of function. This research though, was conducted on children and did not look specifically at verbal fluency performance.
Pai, and Tsaii, (2005) conducted a study on the effects of educational level on the cognitive decline after
onset of Epilepsy. The study was conducted on 64 patients with Epilepsy with no differences in gender or
Epilepsy severity found that patients with epilepsy a high educational level had better cognitive functioning
when tested after the 12th month follow-up. In the area of verbal fluency though, the study found that
subjects with a low Educational level showed improvement. The study though did not use the Controlled
Oral Word Association Test and the Category Fluency test to assess the verbal fluency levels. It also did not
concentrate mainly on verbal fluency but found its focus on the cognitive domains on attention and memory.
Galanopoulou (2008) formulated a framework for explaining sex differences in the brain and behavior relationships in individuals with Epilepsy. These sex differences in the neurobiology of Epilepsy are apparent as seen in the generalization of seizures in men and women, in which more women than men are diagnosed with generalized Epilepsy but localization-related Epilepsies are more frequent in men (Janszky et al, 2004). Since, most cognitive impairments are associated with partial seizures rather than generalized seizures; it may be surmised that women may fair better in cognitive tasks.
Sex differences in verbal fluency are a highly controversial issue in which most studies have shown no differences while a few papers have only shown a slight female advantage. This was seen in a research conducted by Mathuranath et al (2010) who found that sex had no effects on verbal fluency. The research conducted on 153 cognitively unimpaired subjects using both semantic and letter fluency tests showed that sex did not have an impact on verbal fluency. In contrast, a research done by Scheuringer et al (2018) [9] conducted on 19 women and 23 men with similar ages and educational backgrounds in which all participants performed phonemic and semantic verbal fluency tests showed that sex differences in favor of females were only seen when instructions on the test where not neutral. On neutral instructions, sex differences on the test scores were not seen. However this research focused on seeing how a change in instructions could impact sex in verbal fluency. The research also did not use the COWAT or FAS to analyze the verbal fluency levels and both tests did not look at the verbal fluency levels in individuals with Epilepsy. Very few published researches have delved into figuring out how the sex of a person with Epilepsy can impact their levels of verbal fluency. This paper will therefore looked at the issue of sex in its discussion.
Material and Methods
This research was carried out in Lusaka at University Teaching Hospital. The participants were men and
women aged 18 to 50 years of age from UTH. The sample size was 32 participants. Systematic sampling was
used to select participants into the study.
The study was quantitative in nature and employed a cross-sectional survey design. Data was collected through psychological tests: Category Fluency Test (FAS) and Controlled Oral Association Test (COWAT). Data was analyzed using SPSS version 26 and the relationship between variables were tested by use of bivariate analysis.
Results
One of the characteristic of the respondents with epilepsy included the age of onset of epilepsy. The results
revealed that 3 respondents (18.8%) indicated 30 years as the onset of the illness. The other respondent
(6.3%) indicated 59 years as the age of onset of the illness. Two respondents (12.6%) indicated that their
illness started at 4 and 6 months respectively. Other respondents indicated the following years 4 years, 6
years, 12 years, 13 years, 14 years, 16 years, 18 years, 26 years, 27 years, 38 years, and 50 years as the onset of
the illness, each being represented by 6.3%. The results revealed that 25% of the respondents indicated that
their illness lasted for less than one year while the other 25% said that their illness lasted from 1 to 5 years.
One respondent (6.3%) reported that their illness lasted from 6 to 10 years.
The results of the Controlled Oral Word Association Test revealed the following:
Most respondents with epilepsy (63%) were able to say 4 to 6 words. There was only one (6%) non-epileptic
respondent who said 4 to 6 words. There were fewer respondents (3) representing 18% of the epileptic group
who said 7 to 9 words starting with the letter F compared to 7 respondents (44%) from the non-epileptic
group who were able to say 7 to 9 letters. From this analysis, it can be concluded that respondents with
epilepsy had more difficulties in writing words starting with the letter F than respondents who were nonepileptic.
There were 10 respondents (63%) from the epileptic group who said 1 to 3 words using the letter A as
compared to 3 (19%) from the non-epileptic group. Further, more respondents, 11 (69%) from the nonepileptic
group said more words using letter A than 5 (31%) respondents from the epileptic group. Moreover,
12% (2) of the respondents from the non-epileptic group said more than 7 words using the letter A compared
to 6% from the epileptic group. This analysis shows that respondents with epilepsy had a lot of challenges in
writing words starting with the letter A. epilepsy affected them negatively in this part.
In the other tasks, respondents were asked to say any number of words starting with the letter S. Of the
16 respondents (epileptic) who participated in the study, 7 (43.8%) compared to 5 respondents (31%) who
were non-epileptic were able to say 7 to 9 words. The respondents from the epileptic group were not able to
say more than 9 words when compared to the non-epileptic group (19%). From the two groups (epileptic
and none-epileptic) 5 respondents were able to say 5 words using the letter S. there were only 3 (19%)
respondents who were none epileptic who said 1 to 3 words compared to 4 (25%) respondents with epilepsy
who managed to say 1 to 3 words using the letter S.
With regard to words with F Perseverations, 75 per cent (12) of the respondents with epilepsy did not repeat
a word starting with a letter F while 81% (13) respondents without epilepsy did not repeat words with letter
F. The other respondents with epilepsy (25%) repeated one word, while only 12% of respondents without
epilepsy repeated two words starting with letter F. 6% of the respondents did also repeat one word.
The respondents were further asked to say words starting with the letter A. The results showed that 6, (56%)
of the respondents with epilepsy (9) did not repeat any words starting with the letter A. The results were
different for respondents who were non-epileptic. These respondents were represented by 94% (15). These
did not repeat any word. 25% of the respondents with epilepsy did repeat one word whereas respondents
without epilepsy only one (6%) repeated one word. The other 18.7% (3) of the respondents with epilepsy
only repeated two words.
From the table, 10 respondents (63%) with epilepsy did not say any intrusive words starting with the letter
F while 15 respondents (94%) did also not say any intrusive words starting with F. There were 4 respondents
(25%) with epilepsy who said only one intrusive word. Regarding intrusion with two words, there was one
respondent from each group who had an intrusion with two words. Only one respondent with epilepsy had
three intrusive words starting with the letter F. The rest of the results are summarised in table 1.
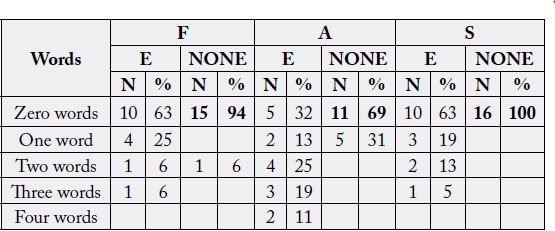
The respondents in this section where asked to state any number of words starting with the letters F, A, and
S. The results are summarised in table 4.8. 88 per cent of the respondents with epilepsy (14) did not deviate
from words starting with the letter F as compared to 75% of respondents without epilepsy (12). 12 per cent,
(2) respondents with epilepsy only deviated with one word while 25% (4) of respondents without epilepsy
also deviated with one word starting with the letter F.
With letter A Variant, 100 per cent of the respondents with epilepsy (16) did not variant from the words starting with the letter A compared to 15 (94%) respondents without epilepsy. One respondent without epilepsy did deviate from the word starting with the letter A. With regard to S Variant, 81 per cent of the respondents with epilepsy (13) did not deviate from any words with letter S while 16 respondents without epilepsy also did not deviated. 13% per cent of the epileptic respondents did variant on one word starting with letter S. 6.3 per cent of the respondents said two words
The adults who participated in the study were tested on verbal fluency performance by the use of Category
Fluency Test (FAS). The results are shown below.
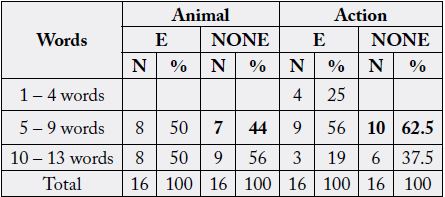
The table above shows that 50 per cent of the respondents were able to say about 5 - 9 words of particular animals and the other 50 per cent managed to say about 10 to 13 words indicating different animals. To surmise, epilepsy did have an impact on the respondents when it came to saying the names of animals.
Table 3 shows the responses of respondents on perseverations and intrusions. On perseverations, specifically
when saying animals, 9 out 16 respondents with epilepsy did not repeat any words while those without
epilepsy who did not repeat any words were 11. Respondents with epilepsy who repeated one word were
6(38%) while those without epilepsy were 4(25%). The rest of the results are shown in the table.
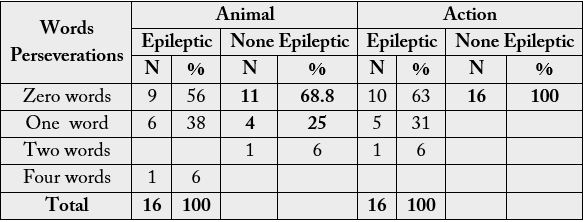
In this part of the Category Fluency Test, respondents were asked to name action words. Both the
experimental and control group responses are summarised. 13 respondents with epilepsy were able to say
names of animals without intrusion. The non-epileptic group had more respondents (15) who did not have
any intrusions among the names of animals. Respondents were also found to have intrusions of one word
in the animal category: 2 respondents with epilepsy and 1 respondent without epilepsy respectively. Two
respondents with epilepsy were able have two intrusive animal names.
With regard to action, 11 respondents with epilepsy did not have problems with saying action words. Those from non-epileptic group who did not have problems with action words were 14. There wasn’t much difference between these two groups. However, there were an equal number of respondents from the two groups who had problems saying action words: one respondent from each group had confusion with one word.
In this part of the study, a description of the association between verbal fluency scores and the clinical
characteristics of Epilepsy (that is the age of onset and duration of the illness) were made.
In order to test the relationship between duration of sickness and verbal fluency scores, a test of correlation
was performed and the results are described below.
i. There was a weak negative none significant relationship between duration of sickness and verbal fluency
score of letter F (r = - 0.346, P = 0.189).
ii. There was a weak positive none significant relationship between duration of sickness and verbal fluency
score of letter A (r = 0.401, P = 0.124).
iii. There was a moderate negative none significant correlation between duration of sickness and verbal
fluency score letter S (r = -0.401, P = 0.124).
i. There was no weak negative none significant relationship between duration of sickness and verbal fluency
score of letter F (r = -0.235, P = 0.380).
ii. There was a weak positive none significant relationship between duration of sickness and verbal fluency
score letter A (r = 0.047, P = 0.864).
iii. There was no statistically significant correlation between duration of sickness and verbal fluency score of
letter S (r = - 0.457, P = 0.14).
In this part of the study, the correlation between duration of sickness and intrusion was performed and the
results described below.
i. There was a weak negative none significant relationship between duration of sickness and verbal fluency
score of letter F (r = - 0.383, p = 0.14).
ii. There was a weak negative none significant relationship between duration of sickness and letter A (r
=-0.314, p = 0.24).
iii. There was a weak positive none significant relationship between duration of sickness and letter S (r (16)
= 0.133, p = 0.62).
Correlations between duration of sickness and variant were performed. The results are described below.
i. There was a weak negative none statistically significant relationship between duration of sickness and
verbal fluency score of letter F (r (16) = - 0.418, p = 0.107).
ii. There was a weak negative none statistically relationship between duration of sickness and verbal fluency
score letter S (r = -0.026, p = 0.924).
In this part of the study, respondents with epilepsy were asked to say the names of animals and some actions.
The correlation results are described below:
i. There was a weak negative none statistically significant relationship between duration of sickness and
Category Fluency Test animal correct (r = - 0.400, p = 0125 which is greater than 0.05).
ii. There was a weak negative none significance correlation between duration of sickness and verbal fluency
score Action correct (r = -0.072, p = 0.791).
iii. There was a weak negative none statistically significant relationship between duration of sickness and
verbal fluency score Animal Perseveration (r = - 0.169, p = 0.532).
From the analysis above, all the correlations were negative and were not statistically significant. In general, the results suggest that the correlation between duration of sickness and verbal fluency scores was negative.
In this part of the study, an attempt was made to find out the relationship between age of onset of epilepsy
and the performance on verbal fluency scores. A correlation involving bivariate analysis was used. The results
are presented below.
i. There was a weak positive none significant relationship between age of onset of epilepsy and verbal fluency
scores for letter F correct (r = 0.253, p = 0.345).
ii. There was a weak negative none significant relationship between age of onset of epilepsy and performance
on verbal fluency scores of letter A correct (r = -0.067, p = 0.806).
iii. There was a moderate positive none statistically significant relationship between age of onset of epilepsy
and performance of verbal fluency score for letter S correct.
i. There was a moderate positive none significant relationship between age of onset of epilepsy and
performance on verbal fluency score for letter F Perseverations (r = 0.448, p = 0.082).
ii. There was a weak positive none significant relationship between age of onset of epilepsy and performance
of verbal fluency score for letter A perseverations (r = 0.195, p = 0.468).
iii. There was a moderate positive none statistically significant relationship between age of onset of epilepsy
and performance of verbal fluency score for letter S perseverations (r = 0.373, p = 0.155).
i. There was a weak positive none statistically significant relationship between age of onset of epilepsy and
performance of verbal fluency scores for letter F Intrusion (r = 0.212, p = 0.430).
ii. There was a strong positive statistically significant relationship between age of onset of epilepsy and
performance on verbal fluency score for letter A Intrusion (r = 0.629, p = 0.009).
iii. There was a weak negative none statistically significant relationship between age of onset of epilepsy and
performance on verbal fluency scores for letter S Intrusion (r = -0.126, p = 0.642).
i. There was a moderate positive none statistically significant relationship between age of onset of epilepsy
and performance of verbal fluency score for letter F Variant (r = 0.341, p = 0.196).
ii. There was a weak positive none statistically significant relationship between age of onset and performance
on verbal fluency scores for letter S Variant (r = 0.205, p = 0.445).
i. There was a moderate positive none statistically significant relationship between age of onset of epilepsy
and performance on Category Fluency Test (Animal) for letter F correct (r = 0.405, p = 0.120).
ii. There was a weak negative none statistically significant relationship between age of onset of epilepsy and
performance on Category Fluency Test for Action Correct (r = -0.194, p = 0.472).
There was a weak positive none statistically significance relationship between age of onset of epilepsy and performance on Category Fluency Test Animal Perseverations (r = 0.010, p = 0.91).
i. There was a weak positive none significant correlation between sex of respondent and verbal fluency scores
of letter F (r = 0.079, p = 0.770).
ii. There was a weak positive statistically significant relationship between sex of respondent and verbal fluency
score of letter A (r = 0.0513, p = 0.042).
iii. There was a weak negative none statistically relationship between sex of respondent and verbal fluency
scores of letter S (r = - 0.077, p = 0.776).
The correlations for respondents without epilepsy were analysed and results are presented below.
i. There was a weak negative none significant relationship between sex of respondent and performance on
verbal fluency scores for letter F Correct (r = -0.103, p = 0.705).
ii. There was a weak positive none significant relationship between sex of respondent and performance on
verbal fluency scores for letter A Correct (r = 0.113, p = 0.678).
iii. There was a weak negative none significant relationship between sex of respondent and performance on
verbal fluency scores for letter S Correct (r = -0.125, p = 0.645).
From the analysis of the two groups above, it can be concluded that the relationship between sex of respondent and performance on verbal fluency scores is weak and were all none significant.
i. There was a weak positive none significant relationship between sex of respondent and verbal fluency
scores of letter F (r (16) = 0.289, p = 0.278).
ii. There was a weak positive none statistically significant relationship between sex of respondent and verbal
fluency scores of letter A (r = 0.160, p = 0.554).
iii. There was a weak positive none statistically significant relationship between sex of respondent and verbal
fluency scores of letter S (r = 0.160, p = 0.554).
With regard none epileptic respondents, the correlation analysis of Sex and verbal scores on Perseveration results are shown below:
i. There was a weak positive none significant relationship between sex and respondent’s performance on
verbal fluency scores on letter F Perseveration (r = 0.224, p = 0.405).
ii. There was a weak positive none significant relationship between sex of respondent and performance on
verbal fluency scores for letter A Perseverations (r = 0.258, p = 0.334).
iii. There was weak negative none significant relationship between sex of respondent and performance on
verbal fluency scores for letter S Perseverations (r = -0.125, p = 0.334).
From the analysis above, it can be deduced that weak relationships exist between sex of respondents and verbal fluency scores on Perseverations. The scoring on verbal fluency scores is not influenced by the sex of the respondents who is epileptic or not.
i. There was a weak negative none statistically significant relationship between sex of respondent and verbal
fluency scores of letter F (r = - 0.072, p = 0.790).
ii. There was weak positive none statistically significant relationship between sex of respondent and verbal
fluency scores of letter A (r = 0.134, p = 0.621).
iii. There was a weak positive none statistically significant relationship between sex of respondent and verbal
fluency scores of letter S (r = 0.135, and p = 0.619).
The scores on verbal fluency scores for respondents without epilepsy are shown below.
i. There was a weak negative none statistically significant relationship between sex of respondent and
performance on verbal fluency scores for letter F Intrusion (r = -0.258, p = 0.334).
ii. There was a weak negative none statistically significant relationship between sex of respondent and
performance on verbal fluency scores for letter A Intrusion (r = -0.135, p = 0.619).
Looking at the above results, most of the correlations for epileptic respondents were positive compared to the correlations for non-epileptic respondents. Therefore sex of the respondents with Epilepsy did not affect their verbal fluency performance.
i. There was a weak positive none statistically significant relationship between sex of respondent and verbal
fluency scores of letter F (r = 0.000, p = 1.00).
ii. There was a moderate positive none statistically significant relationship between sex of respondent and
verbal fluency scores of letter S (r = 0.447, p = 0.082).
Correlations were also performed on respondents without epilepsy and the results are shown below.
i. There was a weak positive none significant relationship between sex of respondent and performance on
verbal fluency scores for letter F Variant (r = 0.289, p = 0.278).
ii. There was a weak positive none statistically significant relationship between sex of respondent and
performance on verbal fluency scores for letter A Variant (r = 0.258, p = 0.334).
In this section, correlation analyses were used to examine the relationship between the sex of respondents
and Category Fluency Test - Animal and Action. The results are described below.
i. There was a weak positive none statistically significant relationship between sex of respondent and verbal
fluency scores- Animal Correct (r = 0.103, p = 0.705)
ii. There was a weak negative none statistically significant relationship between sex of respondent and verbal
fluency scores - Action Correct (r = - 0.095, p = 0.727).
iii. There was a moderate positive none statistically significant relationship between sex of respondent and
verbal fluency scores of Animal Perseverations (r = 0.252, p = 0.346).
iv. There was a moderate positive none statistically significant relationship between sex of respondent and
verbal fluency scores - Action Perseverations (r = 0.308, p = 0.246).
Correlations were also calculated for respondents without epilepsy. The results are presented below:
i. There was a weak negative none statistically significant relationship between sex of respondent and
performance on Category Fluency Scores for Animal Correct (r = -0.378, p = 0.149).
ii. There was a weak positive none statistically significant relationship between sex of respondent and
performance on verbal fluency scores for Action Correct (r = 0.00, p = 1.00).
iii. There was a weak positive none statistically significant relationship between sex of respondent and
performance on Category fluency scores for Animal Perseveration Correct (r = 0.00, p = 1.00).
iv. There was a weak negative none significant relationship between sex of respondent and performance on
Category Fluency Score for Animal Intrusion (r = -0.258, p = 0.334).
v. There was a weak negative none statistically significant relationship between sex of respondent and
performance on Category Fluency Scores for Action Intrusion (r = -0.167, p = 0.537).
Looking at the results for the two groups, it can be concluded that the respondents with epilepsy were able to perform better than the respondents without epilepsy. Most of the responses for respondents with epilepsy were weak positive while for those without epilepsy, responses were mostly negative.
i. There was a weak positive none statistically significant relationship between years of study and verbal
fluency scores of letter F Correct (r = 0.216, p = 0.422).
ii. There was a weak negative none statistically significant correlation between years of study and verbal
fluency scores of letter A Correct (r = - 0.148, p = 0.585).
iii. There was a weak positive none statistically significant relationship between years of study and verbal
fluency scores of letter S (r = 0.136, p = 0.615).
Correlations were also performed for respondents without epilepsy. The results are presented below.
i. There was a weak positive none statistically significant relationship between years of study and performance
on verbal fluency scores for letter F Correct (r = 0.016, p = 0.952).
ii. There was a moderate positive none statistically significant relationship between years of study and
performance on verbal fluency scores for letter A Correct (r = 0.306, p = 0.249).
iii. There was a weak positive none statistically significant relationship between years of study and performance
on verbal fluency scores for letter S Correct (r = 0.080, p = 0.768).
For the two groups (epileptic and non-epileptic) the relationship between years of study and performance on verbal fluency scores was weak.
Years of Study and FAS Verbal Fluency Scores - Perseverations
i. There was a moderate positive none statistically significant correlation between years of study and verbal
fluency scores of letter F Perseverations (r = 0.462, p = 0.071).
ii. There was a weak positive none statistically significant relationship between years of study and verbal
fluency scores of letter A (r = 0.333, p = 0.207).
iii. There was a weak positive none statistically significant relationship between years of study and verbal
fluency scores of letter S (r = 0.077, p = 0.777).
Correlations for respondents without epilepsy were also performed. The results are presented below.
i. There was a weak positive none significant relationship between years of study and performance on verbal
fluency scores for letter F Perseverations (r = 0.072, p = 0.792).
ii. There was a weak positive none significant relationship between years of study and performance on verbal
fluency scores for letter A Perseverations (r = 0.289, p = 0.277).
iii. There was a weak positive none significant relationship between years of study and performance on verbal
fluency scores for letter S Perseverations (r = 0.289, p = 0.277).
From the analysis above, it can be deduced that the relationships between years of study and performance on verbal fluency scores for letters F, A and S Perseverations were weak.
i. There was a weak negative none statistically significant relationship between years of study and verbal
fluency scores of letter F Intrusion (r = - 0.081, p = 0.765).
ii. There was a weak positive none statistically significant relationship between years of study and verbal
fluency of letter of A Intrusion (r = 0.193, p = 0.474).
iii. There was a weak positive none statistically significant relationship between years of study and verbal
fluency scores of letter S Intrusion (r = 0.022, p = 0.937).
Correlations analyses were also performed for respondents without epilepsy. The results are presented below.
i. There was a weak negative none significant relation between years of study and performance on verbal
fluency scores for letter F Intrusion (r = -0.372, p = 0.156).
ii. There was a weak negative none significant relationship between years of study and performance on verbal
fluency scores letter A Intrusion (r = -0.108, p = 0.691).
There wasn’t much difference in the scoring on verbal fluency scores for both epileptic and none epileptic groups. Most of the relationships between years of study and performance on verbal fluency scores for none epileptic group were weak and negative unlike the ones for epileptic group which were weak and positive.
i. There was a weak negative none statistically significant relationship between years of study and verbal
fluency scores of letter F Variant (r = -0.182, p = 0.501).
ii. There was a weak negative none statistically significant relationship between years of study and verbal
fluency scores of letter S (r = -0.072, p = 0.792).
As for the non-epileptic group, the correlations are presented below.
i. There was a weak positive none statistically significant relationship between years of study and performance
on verbal fluency scores for letter F Variant (r = 0.277, p = 0.298).
ii. There was a weak positive none significant relationship between years of study and performance on verbal
fluency scores for letter A Variant (r = 0.289, p = 0.277).
The difference in the performance on verbal fluency scores for both epileptic and non-epileptic groups was mainly on the correlations being negative and positive. For epileptic respondents, the correlations were weak and negative while for non-epileptic group they were weak and positive.
i. There was a weak positive none statistically significant relationship between years of study and verbal
fluency scores of Animal Correct (r = 0.279, p = 0.275).
ii. There was a weak positive none statistically significant relationship between years of study and verbal
fluency scores of Action Correct (r = 0.106, p = 0.695).
iii. There was a weak negative none statistically significant relationship between years of study and verbal
fluency score of Animal Perseveration (r = -0.222, p = 0.409).
With regard to Category Fluency Test for both Animal and Action Trial, the responses for non-epileptic group are presented below.
i. There was a moderate positive none significant relationship between years of study and performance on
Category Fluency Test for Animal Correct (r = 0.303, p = 0.255).
ii. There was a weak positive significant relationship between years of study and performance on Category
Fluency Test for Action Trial (r = 0.537, p = 0.032).
iii. There was a moderate positive none significant relationship between years of study and performance on
Category Fluency Test for Animal Trial Preservations (r = 0.301, p = 0.258).
The analysis of the above findings shows that being epileptic did not largely affect the performance of the participants on Category Fluency Test for both Animal and Action although some impact was seen by some errors. This has been shown by the weak negative and positive correlations for both groups (epileptic and none epileptic).
Discussion of Results
With regard to letter F Correct, the results showed that respondents with epilepsy had more challenges saying correct words that start with letter F than respondents without epilepsy. With letter A, respondents with epilepsy were able to say 6 words correctly. However, respondents without epilepsy said more than six words correctly starting with the letter A. This proved to be a problem with respondents with epilepsy. Respondents were also asked to say words starting with the letter S correctly. The results showed that respondents with epilepsy said 9 words and below. Respondents from the other group (non-epileptic) were able to say more than 9 words with the letter S correctly.
From the results above, it is clear that respondents with epilepsy had problems with saying words starting with the letters F, A and S correctly when compared to respondents from the non-epileptic group. The findings matched that of Thompson and Duncan (2005) who also found that the Epileptic patients assessed performed poorly in phonemic fluency on the ‘F’; ‘A’ and ‘S’ tests. One of the most distinct aspects found that matched with the Thompson and Duncan research was the fact that the least performance on the tests was seen on the ‘A’ aspect of the phonemic test. The reason for this maybe the one highlighted at Scribbeni (2018) where it mentions that ‘A’ is one of the least common phonemes in the English language falling at a number 25 in the total list of 40 phonemes making it more difficult to access from a phonetic store; especially by someone with Epilepsy.
With regard to perseverations, the majority of respondents (75%) with epilepsy did not repeat a word
starting with the letter F as compared to 81% of respondents without epilepsy who did not repeat words
with letter F. Further, the results show that more than half, (56%) of respondents with epilepsy did not
repeat words starting with letter A, compared to 94% of the respondents without epilepsy who did not
repeat words starting with letter A. As before, it still appears that respondents with epilepsy had more
problems with perseveration when saying words with the letter A. It can therefore be concluded that, overall,
respondents with epilepsy had more challenges with repetition of words than respondents without epilepsy;
with more challenges being seen in the ‘A’ and ‘S’ tasks. Buschmann and Schulze-Bonhage (2014) [6], in
their research also found more problems in perseveration in participants with Epilepsy and that seems to tie
in with the results found in this paper. Information found at Dangeuecan et al (2017) [10] which intones
that many research papers have found evidence to suggest that working memory deficits present a significant
barrier to living and learning in many children and adults with epilepsy, seems to provide a reason as to why
perseveration levels were high in individuals with epilepsy.
With regards to intrusion, when saying words starting with letters F, A, and S, respondents with epilepsy
had more challenges than respondents without epilepsy. For instance; more than half, 11 participants, with
epilepsy had intrusion errors in the ‘A, task and 6 participants each in the ‘F’ and ‘S’ tasks. Once again, the
study has shown that respondents with epilepsy will be affected by intrusive errors during verbal fluency
tasks. In phonemic verbal fluency, intrusion errors refer to bringing up words in a verbal fluency task that
do not belong to the specified category. According to Galverna et al (2016) [11], verbal intrusions may be
indicative of decreased inhibition. It has been found that during certain epileptic seizures, there are marked
increases in excitation and decreases in inhibition resulting in a cascade of effects that occur during and
after the seizure which induce both morphological and functional changes in the brain [11]. The inhibition
caused by the seizures could be a factor in explaining why participants with Epilepsy had a number of
intrusive errors.
With variant, the results showed that respondents with epilepsy had no problems with letter F. 80% of
respondents with epilepsy did not have variation errors when saying words starting with letter F, A, and S.
The results simply show that there wasn’t much difference with respondents without epilepsy and Epilepsy
therefore did not have a negative impact on the respondents.
The results showed that 50 per cent of those with Epilepsy as compared to a 56% of those without Epilepsy
managed to say about 10 to 13 names of different animals correctly. The other part which had to do with
action had only 19 per cent of those with Epilepsy as compared to a 37.5% of those without Epilepsy
saying 10 to 13 action words. The explanation is that epilepsy did have an impact on the respondents when
it came to the verbal task. It is further important to note that when comparing the correct responses of the
phonemic fluency test (COWAT) to that of the semantic fluency test (Category Fluency); respondents
with Epilepsy fared better on the semantic fluency test. This is because the category fluency task resembles
everyday production tasks that a person normally involves themselves in, such as creating a shopping list,
making it easier for an individual to access already existing links between related concepts. On the other
hand, the FAS, letter task requires one to access their phonemic categories, categories which are not usually
accessed on a daily basis, (Luo et al, 2010; Katzev et al, 2013).
On perseverations, specifically in the animal task, on the largest number of words (4) persevered by 1
individual with Epilepsy, no individual was found to have persevered from the non-epileptic group. 6
individuals with Epilepsy persevered on 1 word while 4 without Epilepsy persevered also on one word.
There was a slightly higher margin between respondents with Epilepsy and those without Epilepsy on the
action task. 6 respondents with Epilepsy persevered on the action task while no one from the none-epileptic
group persevered. This result shows that a few more respondents with epilepsy had difficulties in saying the
names of animals while even more respondents with Epilepsy had problem with the action task that those
without Epilepsy.
In this part of the Category Fluency Test, respondents were asked to name animal words. The study found
that 13 respondents with epilepsy who were able to say the names of animals without intrusion while
the other 3 had problems. This was slightly different from the respondents without epilepsy of which the
majority, 15, were able to complete the task without intrusive errors. This again shows that respondents with
epilepsy had minor issues when it came to naming animals. It still remains that respondents with epilepsy
had difficulties to say action words.
The overall poor performance by respondents with Epilepsy in the Category Fluency Test is similar to that seen in the research by Thomas and Duncan (2015) and can be explained firstly by (Chertkow and Bub, 2000) which states that the higher order mental strategies used for semantic fluency include generating search strategies, deploying the search strategies and then switching to new strategies. Poor performance in semantic fluency tests is therefore usually associated with difficulties in search strategies as well as with the semantic store.
The correlations between duration of sickness and verbal fluency scores were found to be weak (both negative
and positive) and were none significant. This simply means that duration of sickness had no significant
impact on performance on verbal fluency scores. However, the results from this study seem to contradict
those of Elger (2004) who found that prolonged duration of Epilepsy also caused a deterioration of general
intellectual functioning, (Elger, 2004). Elger’ findings are supported by those of Iterson et al (2014), who
found that there was a downward progression of function, from the first test during earlier periods of
Epilepsy to the second test. This shows clearly that the longer a child has the condition, the larger the
likelihood of there being intellectual dysfunction and concurrently language deficits. This discrepancy could
be that Iterson study was conducted in children and was also conducted at intervals to properly aid in
measuring brain performance over time; while this research focused on adults and was conducted in one
specific time period.
The correlations between age of onset and performance on verbal fluency scores were found to be both weak
negative and weak positive. Though weak correlations were found, there seems to be an impact as respondents
with Epilepsy did find challenges in giving correct responses and others had challenges with repeating words
and still others had intrusive errors. These findings are in agreement with those of Sillanppa et al (2015) [12]
who studied the impact of childhood‐onset epilepsy on a variety of outcomes across the life span and found
that many respondents presented with neurologic disorders including verbal communication disorders.
The correlation between the sex of the respondents and performance on verbal fluency scores was found
to be insignificant. The results were similar for respondents without epilepsy. This means that the sex of a
respondent has no impact on the performance on verbal fluency scores. The results of this study agree with
most of the researches, including that of Marunarath (2010) which found that sex has no influence on verbal
fluency. Scheuringer et al (2018) [9] also found that 19 women and 23 men with similar ages and educational
backgrounds in which all participants performed phonemic and semantic verbal fluency tests showed that
sex differences in favor of females were only seen when instructions on the test where not neutral; in this
study though, instructions given to participants in the research were all consistent and neutral.
For both the epileptic and none epileptic group, the correlation results were also found to be none significant.
This means that educational level or years of study did not influence performance on verbal fluency scores.
However, this study differs with the findings of Pai, and Tsaii, (2005), who found that overall, Epilepsy
patients with a high educational level had better cognitive functioning when tested after the 12th month
follow-up. The research conducted by Pai and Tsaii was conducted in China, of which its education system ranks highly, as one of the best in the world, (wenr.wes, 2019). The Zambian education system on the
other hand does not rank nearly as high and it is probable to find inconsistencies such as school-leavers
that cannot read adeptly or speak fluently while having middle-scholars who can read and speak fluently,
(Asimbuyu, 2011) [13-15].
Conclusion
The results have established that patients with epilepsy did not perform well on verbal fluency scores as
compared to their counterparts, the respondents without epilepsy. Patients with epilepsy had problems on
performance on both the COWAT and Category Fluency Test; with challenges giving the correct responses
and with intrusive, perseveration and variant errors being present in their responses. The correlations
between verbal fluency scores and years of study, sex of respondent and duration of sickness were found to
be insignificant. It can be there be concluded that years of study, duration of sickness and sex of respondents
for both epileptic and none epileptic groups did not influence performance on both COWAT and Category
Fluency Test.
Recommendations
Based on the findings of the study, the following recommendations were made:
i. There is need for stakeholders to create neuropsychological assessment programs, to assess executing
functioning and higher order cognitive abilities (switching, working memory, inhibitions, switching,
searching) and verbal fluency, in patients with Epilepsy at clinics and hospitals so as to identify problems
with executive functioning and verbal fluency as early as possible.
ii. There is need for stakeholders to create information programs that educate individuals with Epilepsy
on the effects that their condition can have on their cognitive performance (executive function and verbal
fluency) to help them take preventative precautions such as consistently adhering to their medications.
iii. Stakeholders should also put in place language therapy programs in clinics and hospitals, such as vocabulary
expansion programs and phonological awareness skill workshops to facilitate the acquisition of phonological
and semantic skills to help epileptic patients found with problems in language and verbal fluency.
Bibliography

Hi!
We're here to answer your questions!
Send us a message via Whatsapp, and we'll reply the moment we're available!