Biography
Interests
Shimon Shatzmiller*, Marina Kovaliov, Galina Zats & Inbal Lapidot
Department of Chemical Sciences, Ariel University, Israel
*Correspondence to: Dr. Shimon Shatzmiller, Emeritus Professor, Department of Chemical Sciences, Ariel University, Israel.
Copyright © 2018 Dr. Shimon Shatzmiller, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
The composition deals with the antibacterial situation in contemporary infections treatment given the drugresistant bacteria; the nosocomial pandemic requires novel attitude to combat the killing bacteria. In the center are the antimicrobial peptides and surrogate. Small molecules and polymers.
Arias like soil sterilization and food preservation emerge again because the traditional agents are classified today as toxic or environmentally damaging materials.
The Scientific community is worried regarding the deteriorating situation in the healthcare installation. The Nosocomial pandemic is one of the mail killers of humans, only in Israel (8 million), 6000 pass away due to Hospital-acquired infections, HAI, annually. The two strategies in where the focus on eradication of bacteria- new drugs and hygiene are discussed. The unfortunate situation is that both strategies do not stop the mortality of infected humans caused by the currently developing new strands of microbes.
Effort and strategies for finding new agents are discussed.
The research from the agent’s invention is mainly based on the antimicrobial peptides, the lantibiotics and lactoferricin. The emergence of novel antibacterial agents by antibacterial peptides is discussed.
Abstract
The frequent failure of antibiotic treatment is an acute public health problem. The most apparent reason is that the development of bacterial resistance compromises the successful use of any therapeutic agent. An example is Aminoglycoside antibiotics, such as gentamicin and kanamycin, directly target the ribosome, yet the mechanisms by which these bactericidal drugs induce cell death are not fully understood. The era of antibiotics based on intracellular target components seem to end. Also, the era of antibiotics that hinder cell wall synthesis is losing momentum. The long list of antibiotic drugs to its families is becoming insufficient since it hardly answers the common bacterial infections acquired nonsocial or otherwise. It seems, that exploitation of the Antimicrobial Peptides. AMP, Examples include cecropins, from insects, magainins from amphibians and cathelicidins, from mammals, and their way to eradicate bacteria by cell wall disruption is one of the few clues to the problem of microbe’s resistance, eradication of persister cells, dormant cells and mutants may become a real option and bring remedy to those who suffer the incurable infection caused by those killer bacteria.
The antimicrobial peptides are nature’s weapon to combat the invasion of microbes as host defense
agents, the first line of defense. However, the natural peptides suffer from serious drawbacks
the first in the list is their being peptides and sensitive to enzymatic digestion. The approach of
peptidomimetics [1] bears many advantages over the gift of nature, and that is:
1. Stability in the enzymatic world.
2. The option of human design.
3. Use of un-natural components to the construction of the mimetic architecture.
An approach based on those lines enables the introduction of the novel design of components that may aid in the eradication but are novel for nature. It may allow the exploration of the way of selective eradication of microbes in the presence of beneficial flora.
There are many obstacles in this way: It is needed to learn about the mechanisms in which the AMP disrupt the cell membranes in full detail. Today, many ways were found in which the membranes are destroyed. It was found that low molecular weight (MW = 500) non-peptide mimics can eradicate bacteria, and small structural modification may lead to preference in eradication at the will of either Gram-positive or Gram-negative bacteria. The ruthless selection for resistant bacteria, coupled with an insufficient investment in antibacterial research, has led to a steady decline in the efficacy of existing therapies and a paucity of novel structural classes with which to replace them or complement their use.
In This Chapter, we survey the antibacterial Agents situation with emphasis on the novel trend of developments based on antimicrobial peptides and their surrogates.
The current situation that strikes us with nosocomial infections may bring us back to the times reminiscent of the photo [2].

Opening Words
Antimicrobial agents are some of the most widely, and often injudiciously, used therapeutic drugs worldwide.
A fundamental philosophy of medical treatments, including a combination of antibiotics, is that different
drugs work through different mechanisms and that the results of using multiple drugs will be synergistic
to some extent. Because they have different dose-limiting adverse effects, they can be given together at full
doses in therapy regimens [1].
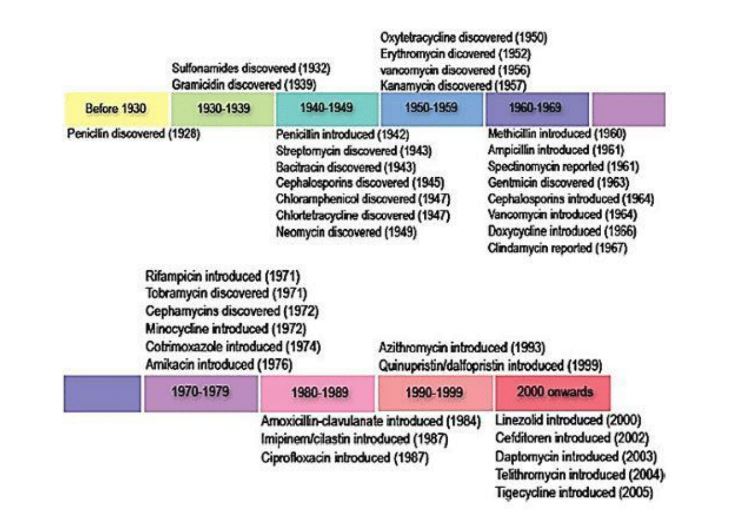
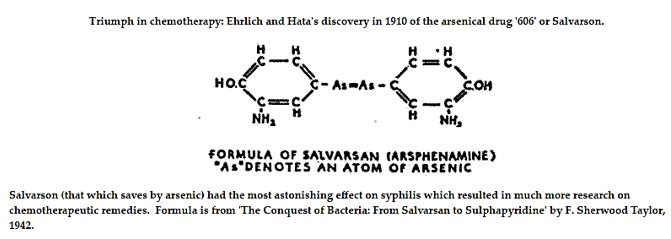
This idea prevailed through the years of infection therapy. In the last 80 years since the first treatment of syphilis with salvarsan by Ehrlich. The initially expressed idea of looking for the “magic bullet” as ideal agent that will cure infections is pursued until now, but the bullet is still elusive. Ehrlich concentrates on work to create his “magic bullets” - chemicals injected into the blood to fight various diseases, thus pioneering antibiotic therapy for infectious diseases (later adopted by others to fight cancer) [3].
Ehrlich and his co-workers tried hundreds of chemicals on the microbes that caused syphilis. In 1909, Ehrlich’s new colleague Sahachiro Hata (1873-1938) brought with him a method of producing syphilis infections in laboratory rabbits, and discovered (1910) that drug no. 606 worked. The first ‘magic bullet’ had been found, and was marketed under the name Salvarsan. This was the first “magic bullet”.
In the last period, of 80 years, with a peak at the period of the years 1960-1980, Human kind enjoyed on the basis of e4hrlich’s findings, a period of triumph over the ancient dwellers. Our nemesis, the most ancient dwellers of this planet: the microbes (archaea, bacteria, fungi, viruses and more).
Since the days of Ehrlich, the development of technology, in chemistry and Biotechnology, provided agents of various nature: those from chemical synthesis [4] and others from bio-synthesis [5]. Materials that could eradicate the broad spectrum of microbes very efficiently and allowed in this short era the extension on human life longevity from 40 years in average to 80 years of today [6]. Humans took this for granted, those were the days that people thought they will never end, not knowing that the “wonder bullet agents” do not eradicate the microbes completely, the few remaining organisms in the margins proliferated, but this time to new unknown races that were resistant to the “wonder agents”. And here we are again today, facing the microbe world without effective weapons in this stage of campaign [7].
One of the major mistakes exercised by the healthcare system was and is still the abuse, over application of the antibiotic drugs. The common and use practically without barrier is one of the main reasons why humankind in now in the grim situation of “fighting without effective weapons” This is today one of the main killers of people and will no doubt sky rocket soon [8].
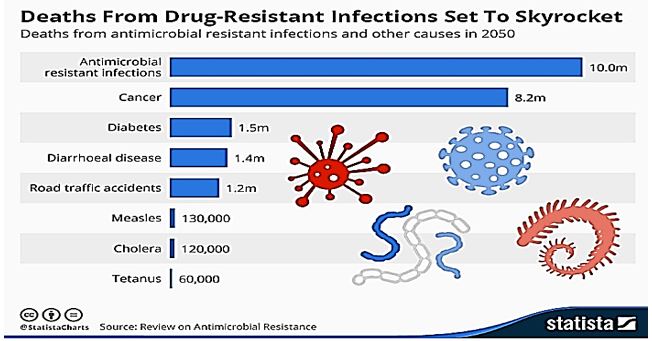
The nosocomial infections are currently on their way to become the major morbidity causing factor in our life. One of every 25 entering a healthcare facility is infected; millions die due to lack of remedy, a novel idea or strategy is badly needed [9]. Antimicrobial peptide may bring remedy in cases of the heavy disease. For example, Burkholderia pseudomallei is a category B agent that causes Melioidosis, an acute and chronic disease with septicaemia [10]. The current treatment regimen is a heavy dose of antibiotics such as ceftazidime (CAZ); however, the risk of a relapse is possible.
Peptide antibiotics are an alternative to classical antibiotics as they exhibit rapid action and are less likely to result in the development of resistance.
Given the grim current situation and the alarming perspectives (see chart above), The effort is spreading in mainly two different strategic option:
1. “Let them become ill; we will cure the disease” - Pharma industry. The pipeline contains:
a) Improving existing drugs,
b) a better understanding of the mechanism of action needed for the design synthesis of DE-NUVO
antimicrobial agents.
c) Isolation of agents from the vast pool of natural products.
d) Mimicking natural bioactive compounds. The design and synthesis of surrogates of bioactive compounds.
2. Prevention of infection – “keeping the microbe-free environment in healthcare facilities”- more effective
sterilization and antiseptic treatment of the whole facility -The Chemical industry.
a) Better disinfecting agents.
b) Better hygiene behaviors.
c) Novel sterilization agents needed to keep the whole installation sterile.
A combination of the two attitude is penetrating, slowly, the healthcare system. Both have the serious drawback that enables the proliferation of the “Nosocomial infections” death pandemic.
Introduction and Overview
The human body is equipped with the immune system created to combat infections microbes. We carry
in our saliva or mother milk. For example, agents (Lactoferrin [11,12]) that eradicate microbes, so do all
creatures living on this planet in their innate immune system, however, the most ancient dwellers of earth.
The microbes (bacteria) can change their genetic material [13] and develop novel races that are indifferent
to these eradicating agents, whatever their molecular structure will be. The genetic transfer of microbes is
a well-studied process and makes the transfer of toxic features from one race of microbes to another [14].
The emergence of the “wonder agents”- the antibiotics. The antibiotics of the early 30-40ies of the last
centuries was a result of an immense technological and scientific breakthrough in human understanding.
Exploration. Theoretical and practical science in many areas. Progress in:
a) The advancement in mechanistic thinking,
b) Novel chemical experimental and theoretical comprehension,
c) Understanding of the photo-physical properties of matter contributed to the preparation of the “Sulfa”
drugs,
d) Biotechnology,
e) The harnessing of microbes for the discovery and manufacture of chemical compounds, the “Penicillins” antibiotics,
f) The establishment of institutes for research in biotechnology, chemistry, and physics in many countries
was an initial “kick” to the findings of antibiotics,
g) Later psycho-pharmaceuticals and developments in the cure of internal-medicine dysfunctions,
h) This was the advent of the modern medicine and industry.
However, some leaks were not handled properly, for example, the eradication of microbes; some microbes were saved from the unimagined fierce attack, they are the ancestors of today’s preliminary stages of the catastrophe.
Metals were applied as antimicrobial agents already in antiquity; the persians used vessels made of copper and silver to avoid corruption of agricultural products. In pre-antibiotic times, mercury was a major agent to cure infections. Today, nanoparticles of metals like gold, silver, copper, magnesium, and metal oxides are applied in large scale as antimicrobials for many uses [15]. However useful, these are not applied to therapy but mostly as antimicrobial agents in fabrics, sterilization of soil or in plastic products. However, their potent antimicrobial efficacy due to the large surface area to volume ratio has provided them an edge over their chemical counterparts which are facing the problems of drug resistance.
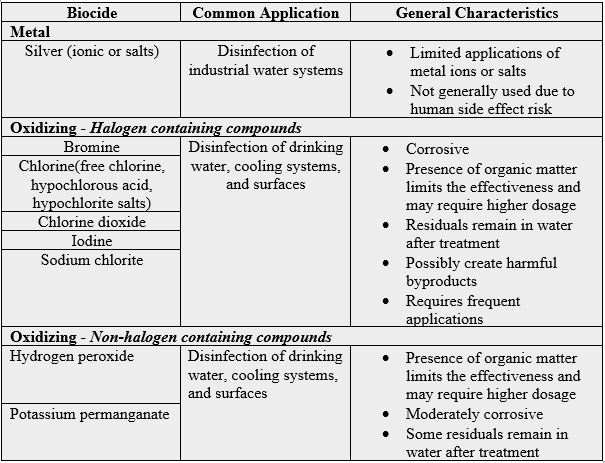
Halogens, main chlorine, are applied as antimicrobial agents in large amount to keep drinking water from microbes [16]. Halogens in Particular Iodine are applied as topical antimicrobial agents in wound healing as elements or in liquid solutions.
In the table 1 below are some inorganic examples of biocides applied for drinking water [9]:
Broad-spectrum antibiotics are powerful fighters with one flaw: unable to smite every bacterium, those immune to their wrath thrive. With every ear infection, we treat, and every healthy cow prophylactically dosed with antibiotics (which also helps fatten the animals), we make these drugs less useful for future generations.
Scientists all over the globe are in a race with evolution, scrambling to understand the underlying mechanisms of antibiotic resistance and to discover new ways to fight bacteria. We must diversify our methods for treating bacterial infections and simultaneously reduce the number of antibiotics we use.
Scientists all over the globe are in a race with evolution, scrambling to understand the underlying mechanisms of antibiotic resistance and to discover new ways to fight bacteria. We must diversify our methods for treating bacterial infections and simultaneously reduce the number of antibiotics we use.
Where could our advances in technology aid us in the pursuit of a novel agent in the combat against microbes?
Humans wandered how animals survive the most polluted environments, yet they are free from infections, some even can regenerate parts of their bodies (tail, leg) that were ripped off in their fightings. Here modern technology helps. Today, we can isolate and determine the structure of the compounds that are enabling the crocodiles and lizards and frogs and other to resist infections and re-assemble their organs. Chemical and Biological laboratory advanced analytical methods can crack every molecular structure, and synthetic methods are available to mimic the biologically active compounds that movies the processes of life. However, we do not fully understand the complex situation in the microbe’s cell to interfere with the high productivity of microbe’s multiplication which we have to stop. A lot is done to penetrate the secretes of the microbe cell membrane. Currently, no clear picture is available to science [17].
In the middle ages, Horrendous pandemics ruled the world and exterminated almost the human race. The back death for example. It was the concept of Hygiene, personal and environmental that saved humankind. At the early age of the 14th century, the following was denounced:
In 1349, Edward III complained to the Lord Mayor of London that the streets of the city were filthy [18]:
“Cause the human faces and other filth lying in the streets and lanes in the city to be removed with all speed to places far distant, so that no greater cause of mortality may arise from such smells.”
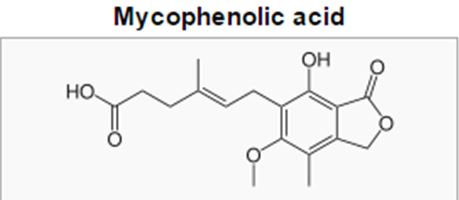
Mycophenolic acid was discovered in 1893 by Bartolomeo Gosio. It was applied as antibiotic drug and was the first antibiotic agent to be synthesized and isolated in the pure form. It was eventually found to be a broadspectrum acting drug having antiviral, antifungal, antibacterial, anticancer, and antipsoriasis properties.
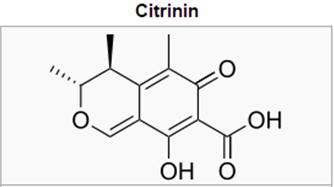
Another disappointment was the agent CITRININ that was developed as antibacterial agent but was found to bwe34 toxic to mammals [19].
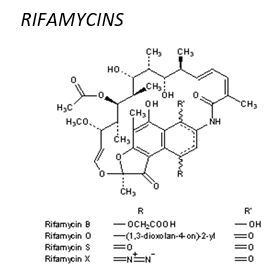
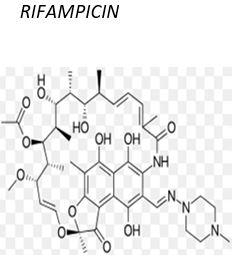
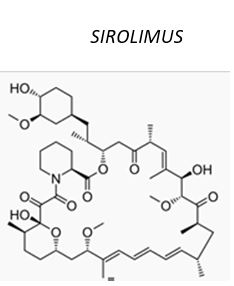
Macrolides related to Rifamycin, Rifampicin, also known as rifampin, is an antibiotic used to treat several types of bacterial infections. This includes tuberculosis, leprosy, and Legionnaire’s disease. It is almost always used along with other antibiotics.
Recently, a high volume of use due to much attention has focused on the potential use of rapamycin (Sirolimus) to prevent cardiac stent restenosis. Most cardiac stent architectures include today Sirolimus as antibiotic built in as an ingredient [20].
The macrolides and ketolides [21] are a class of natural products that consist of a large macrocyclic lactone ring to which one or more deoxy sugars, usually cladinose and desosamine, may be attached. The lactone rings are usually 14-, 15-, or 16-membered. Macrolides belong to the polyketide class of natural products. Some macrolides have antibiotic or antifungal activity and are used as pharmaceutical drugs.
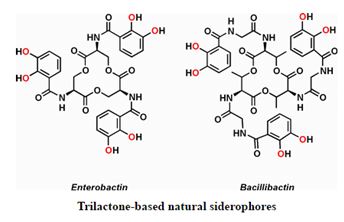
There is an ever standing effort to discover remedy from products of nature from all kingdoms of natural creatures manty families of organic compound were isolated and tested some are iron complexing agents: the siderophores. Bacteria are processing iron in their respiration process for this they Apply The secretion of low-molecular-weight organic chelators called siderophores is one of the main iron mobilizing strategies. Since this is a major maintaining process, Siderophores like enterobactin, bacillobactrin [22] or analogs may be applied as Trojan Horses [23] to introduce effective antibiotics to bacteria cells.
Other compounds types have been identified [24] as potential antibacterial agents: Acetylenes, Coumarins, Flavonoids and isoflavonoids, Iridoids, Lignans, Macrolides, Phenolics (other than flavonoids and lignans), Quinones, Steroidal saponins, Terpenoids, Xanthones, Polypeptides.
Antibacterial agents target various sites in the microbes which stops its life cycle as in the following example (cartoon):
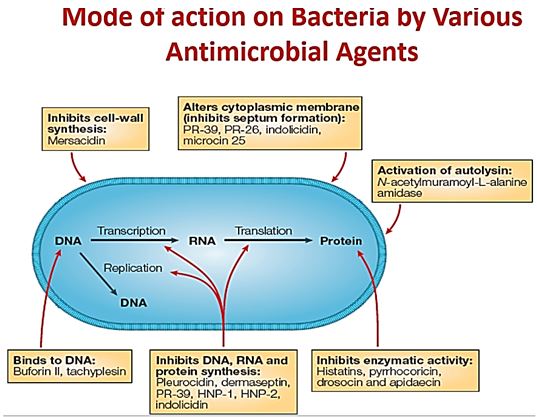
The majority of the antibiotic agent in use today are aiming at the genetic materials (DNA, RNA) but a great portion is effective at the cell membrane domain. The mechanisms of action of today’s agent are clarified to a great extent in particular after the elucidation of the structure of the ribosome [25].
Antimicrobial Peptides Based Agents
The developing pandemic of nosocomial microbial infections is gathering momentum; many die from
the microbial infections acquired in the healthcare facilities. Unfortunately, no remedy is available [26].
Nowadays, the grim situation is taking a toll of hundreds that pass away every day [27]. There is no doubt, a
new approach to the novel design of antimicrobial active and selective drug is needed [28]. The antimicrobial
peptides (AMP), amphipathic structures [29].
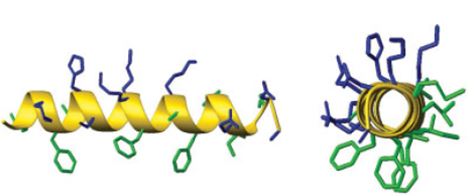
composed of amino acids, the natural first line of the innate immune system, may provide the clue based on the novel mechanism of microbial eradication in which the operate in the microbes [30]: disruption of the cell membranes [31,32]. The compounds eradicate microbes efficiently. These are broad bands lytic biocides. The agents eradicate all sorts of bacteria: Gram positive, Gram negative and mycobacteria in a similar effectivity. However, Since organisms like humans live in symbiosis with many strands of essential flora, this vicious biocide have to be tamed and thereby aid in the cure of the nosocomial proliferating killer bacteria. The Methicillin-Resistant Staphylococcus Aureus (MRSA), Acinetobacter baumannii is a typically short, almost round, rod-shaped (coccobacillus) Gram-negative bacterium, Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumonia, Pseudomonas aeruginosa, and Enterobacter species. All can get infected by Natural Transformation Facilitates Transfer of Transposons, Integrons and Gene Cassettes between Bacterial Species, [33] by genetic transfer (conjugation, transformation, and transduction, horizontal gene transfer [34]) to become Carbapenem Resistant Enterobacteriaceae (CRE). If possible, eradicate only those.
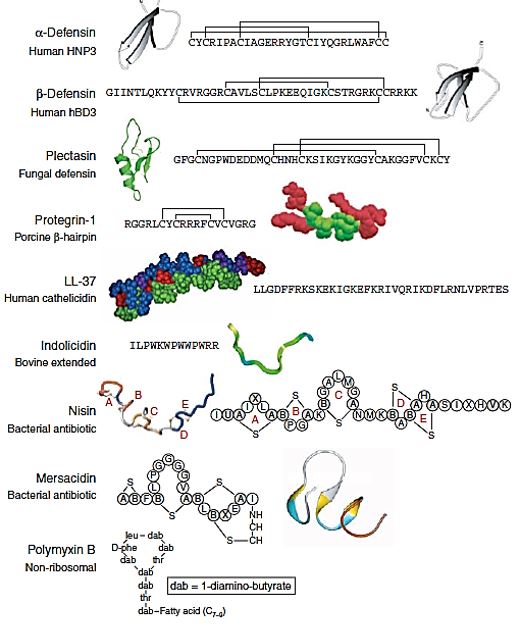
Today, very toxic antibiotic like polymyxin (colistin) [36] are applies in cases of the common sepsis infection that may be the case with the killer bacteria, many patients pass away during this treatment [37]. There is a diversity of AMPs, many originate in insects and show a diversity of biological activities [38]. Insect AMP can selectively inhibit the pathogenic fungus Fusarium graminearum development [39]. However, the general knowledge of AMP classifies them as broad band biocides. The eradicate all microbes.
The natural array of the 2000 AMPs suffers from the following main disadvantages when therapy is
considered:
1. Low oral bioavailability;
2. A short half-life because of their rapid degradation by proteolytic enzymes of the digestive system and
blood plasma;
3. Rapid removal from the circulation by the liver and kidneys;
4. Poor ability to cross physiological barriers because of their general hydrophilicity;
5. high conformational flexibility, sometimes resulting in a lack of selectivity involving interactions with
different receptors or targets (poor specific biodistribution), causing activation of several targets and leading
to side effects; eventual risk of immunogenic effects; and high synthetic and production costs [40].
The most significant useful feature of AMPs is their potent bactericidal activity against multi-drug-resistant bacteria, which is thought to be due to differences in their mechanism of action compared to traditional antibiotics [41]. Resistance to microbial attack can be engineered into organisms (plants) by gene technology based on AMP and hybrid AMP [42]. A single amino acid when conferred to the AMP at the right position, can alter the architecture and the antimicrobial properties of the AMP and with it new recognition properties [43].
All this effort was carried out. The polypeptides research had the most promising results by the identification of the “Antimicrobial peptide” (Antimicrobial peptides are relatively small (6 to 50 amino acids). These are amphipathic molecules of variable length, sequence, and structure with activity against a wide range of microorganisms including bacteria, protozoa, yeast, fungi, viruses and even tumor cells.) as a class of many compounds that exists in all creatures in the innate immune system. People have high expectations relying on these compounds.
For more than 80 years, antibiotics have been nothing short of miraculous. Capable of killing bacteria without killing people, they’ve turned grave illnesses into mere annoyances, providing doctors with a license to shoot first and ask questions later, and do so safely - or so we thought.
Broad-spectrum antibiotics are powerful fighters with one flaw: unable to smite every bacterium, those immune to their wrath thrive. With every ear infection, we treat, and every healthy cow prophylactically dosed with antibiotics (which also helps fatten the animals), we make these drugs less useful for future generations.
Scientists all over the globe are in a race with evolution, scrambling to understand the underlying mechanisms of antibiotic resistance and to discover new ways to fight bacteria. We must diversify our methods for treating bacterial infections and simultaneously reduce the number of antibiotics we use.
There are two main approaches to overcome the microbes issue:
1. Find a new remedy for the sick (New antibiotic research)
2. Prevent the ability of the microbes to infect their targets – the people, Humans (new antiseptic and
Hygiene research).
We will discuss the matter given these two points.
One of the major factors leading to the current loss of the antibiotic capability of modern therapy is the overuse or abuse of antibiotic. In many instances, the application is almost superfluous. Here is what a patient should be aware of when advised to an antibiotic based treatment:
5 Questions to Ask Your Doctor Before You Take Antibiotics
Use these five questions to talk to your doctor about when you need antibiotics–and when you don’t.
Antibiotics can help prevent or treat some infections. But if you use them for the wrong reason, they may
cause unnecessary harm. Talk to your doctor to make sure you only use antibiotics for the right reasons - and
at the right time.®
1. Do I really need antibiotics? Antibiotics fight bacterial infections, like strep throat, whooping cough, and
symptomatic bladder infections. But they don’t fight viruses-like common colds, flu, or most sore throats and
sinus infections. Ask if you have a bacterial infection.
2. What are the risks? Antibiotics can cause diarrhea, vomiting, and more. They can also lead to “antibiotic
resistance”-if you use antibiotics when you don’t need them, they may not work when you do need them.
3. Are there simpler, safer options? Sometimes all you need is rest and plenty of liquid. You can also ask
about antibiotic ointments and drops for conditions like pink eye or swimmer’s ear.
4. How much do they cost? Antibiotics are usually not expensive. But if you take them when you don’t need
them, they may not work for you in the future - and that may cost you a lot of time and money.
5. How do I safely take antibiotics? If your doctor prescribes antibiotics, take them exactly as directed, even
if you feel better.
Antibacterial Agents for Topical Treatments
The application of antibiotics in the clinic is depending on the availability of antibiotic agent of various
families. Topical Antimicrobials for Burn Wound Infection The suggestion is to apply antibiotics from
the following sorts based on Silver, iodine, photodynamic therapy, chitosan, antimicrobial peptides, and
miscellaneous agents [44]. This list shows that antibiotic therapy is complicated and does not rely on
magic bullets. The application is for topical wounds and burns; inner damage might become even more
elusive. Many antibacterial peptides, therefore, preferentially disrupt prokaryotic membranes and eukaryotic
mitochondrial membranes rather than eukaryotic plasma membranes [45].
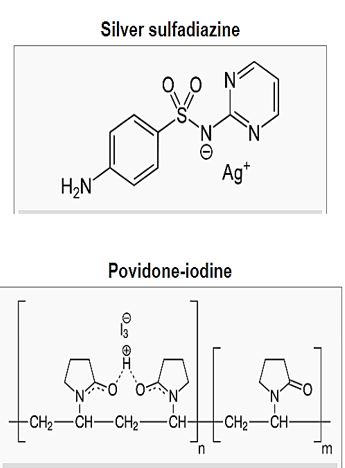
Silvasine, Silver sulfadiazine, sold under the brand Silvadene among others, is a topical antibiotic used in partial thickness and full thickness burns to prevent infection, was shown to be cytotoxic [46]. The Iodine Complex Povidone-iodine is one of the most used solutions to disinfect skin wounds [47]. The imergence of resistant, dormant and persister bacteria makes the antibacterial treatment less effective. In summary, the discovery and development of antibiotics during the 20th century provided potent antimicrobial agents with high specificity, which revolutionized clinical therapy and marked the decline of many former remedies.
However, the relentless emergence of antibiotic resistant strains of pathogens, often with multiple antibiotic resistance, together with the retarded discovery of novel antibiotics has led to the need to find alternative treatments. Faced with the prospect of increased prevalence of antibiotic-resistant pathogens, and the diminished effectiveness of current therapies, careful consideration of treatment options is now important.
The development of antibiotics during the 20th century marked the decline of many former remedies, but the emergence of antibiotic resistant strains of pathogens has led to the need to find alternative treatments.
a) The judicious, prophylactic use of antiseptics may prevent the development of infections that will minimize
antibiotic use, as well as promoting healing.
b) The evidence concerning the efficacy of topical antimicrobial agents in the management of wounds is
confused.
c) Larger better designed trials to assess clinical efficacy and cost implications are necessary.
d) Although reports of resistance are limited, misuse and abuse of antiseptics must be avoided.
The list of antibiotic medication recommended antibiotic treatment for diabetic foot infection is even longer [48]. The proper use of antibiotics in the treatment of the diabetic foot remains contested: one view is to administer antibiotics only in the presence of clinical infection; the other one is to give antibiotics freely to all patients with ulcers [49]. It is an effort that in many instances fails and demands the amputation of the infected leg. Cephalosporins, Penicillins, Fluoroquinolones, Carbapenems, and novel Miscellaneous agents: Aztreonam Clindamycin Piperacillin/clindamycin (iv) (24), Pexiganan (topical) Linezolid (iv or po) Daptomycin (iv).
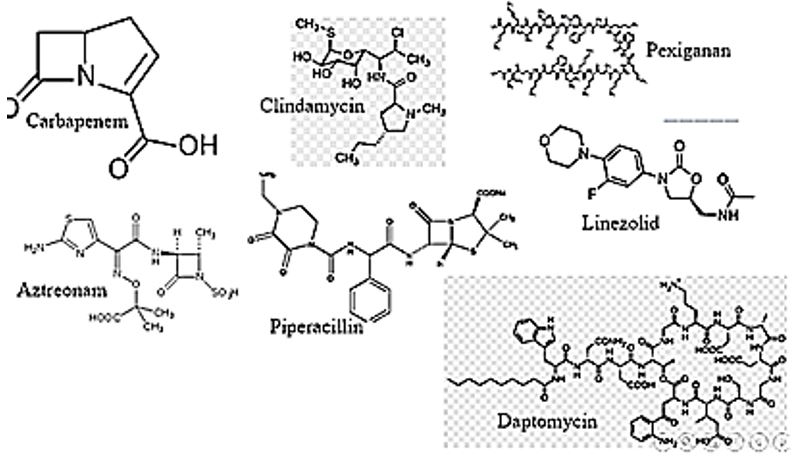
Post-Surgery Infections
The Surgery is carried out in operation halls which are sterilized to prevent as much as possible infections
due to the operation. However, the “lion’s share” min nosocomial infections are acquired due to operations.
A nosocomial infection, also known as a hospital-acquired infection or HAI, is an infection whose development is favored by a hospital environment, such as one acquired by a patient during a hospital visit or one developing among hospital staff. Such infections include fungal and bacterial infections and are aggravated by the reduced resistance of individual patients [50].
In the United States, the Centers for Disease Control and Prevention estimate that roughly 1.7 million hospital-associated infections, from all types of microorganisms, including bacteria, combined, cause or contribute to 99,000 deaths each year. In Europe, where hospital surveys have been conducted, the category of Gram-negative infections are estimated to account for two-thirds of the 25,000 deaths each year. Nosocomial infections can cause severe pneumonia and infections of the urinary tract, bloodstream and other parts of the body. Many types are difficult to attack with antibiotics, and antibiotic resistance is spreading to Gramnegative bacteria that can infect people outside the hospital.
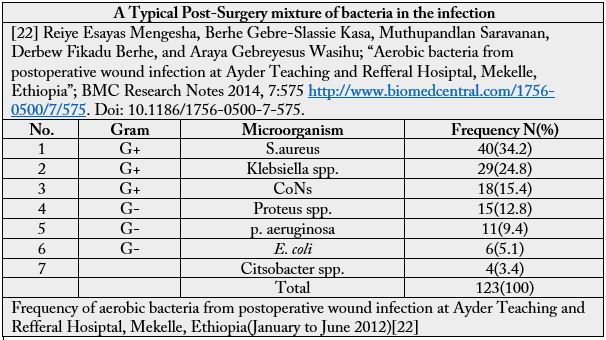
While there are antibiotic drugs that can treat diseases caused by Gram-positive MRSA, there are currently few effective drugs for Acinetobacter.
Here is a long list of antimicrobial agent recommended for surgery treatments [51]; Tobramycin and Vancomycin are mentioned on the long list of agents. Both are effective as killers of Gram positive bacteria, MARSA (methicillin-resistant Staphylococcus aureus) drug agents.
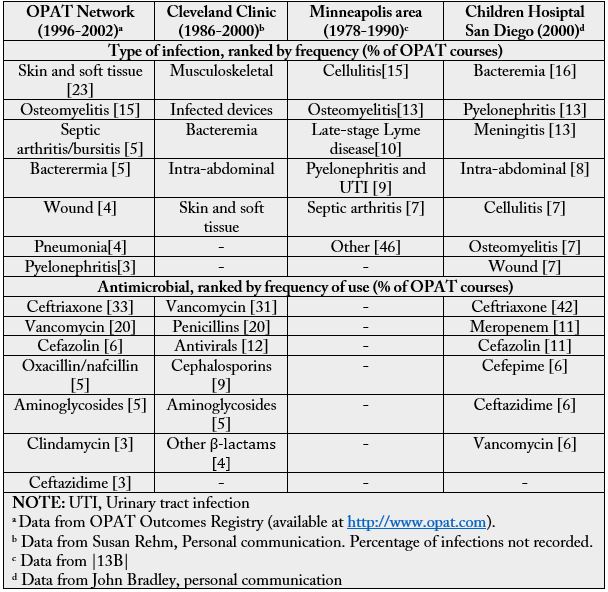
The situation is almost uncurable where there occurs a CRE, Gram-negative bacteria, (carbapenem-resistant Enterobacteriaceae) infection.
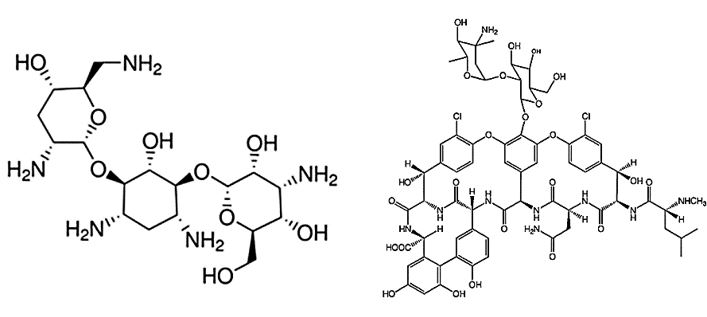
Combating CRE infections is currently a very hard to do. The emergence of carbapenem resistance in Enterobacteriaceae is an important threat to global health. Reported outcomes of infections with carbapenemresistant Enterobacteriaceae (CRE) are poor. Very few options remain for the treatment of these virulent organisms. Antibiotics which are currently in use to treat CRE infections include aminoglycosides (An example is Aminoglycoside antibiotics, such as gentamicin and kanamycin, directly target the ribosome, yet the mechanisms by which these bactericidal drugs induce cell death are not fully understood) [52], polymyxins, tigecycline, fosfomycin, and temocillin. Also, the role of combination therapy, including carbapenem containing regimens, remains to be defined. There are several important concerns regarding all of these treatment options such as limited efficacy, increasing reports of resistance, and specific [53]. As almost no novel agents have been introduced to combat multiple-antibiotic-resistant organisms, clinicians may be forced to rediscover colistin and colistimethate (colistin methanesulphonate sodium, colistin sulphomethate sodium). In the drawing are the few substances that are applied today as antibacterial agents in such circumstance. (Colistin [54], Tigcycline, Fosphamycin, Amicacin and Temocyclin). These are Toxic comound and the application introduces the high morbidity (up to 80% in some reports) [55]. Better agents are needed badly.
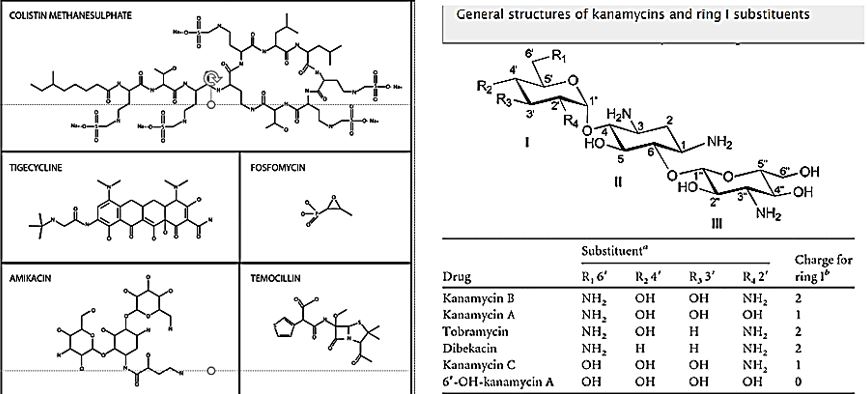
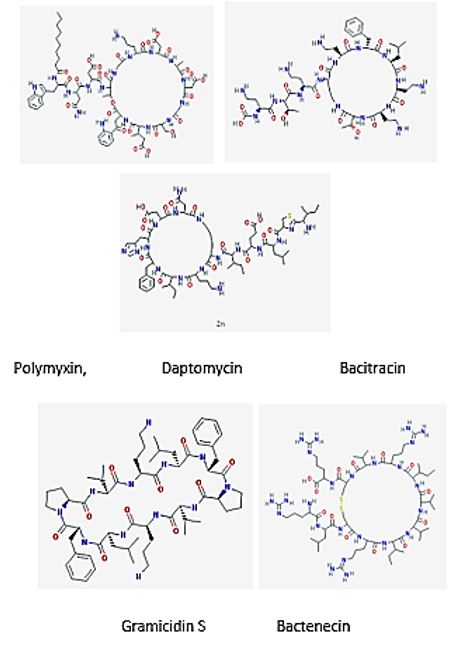
Today, some of the most efficient antimicrobial drugs are based on cyclic peptides, Examples are: Polymyxins, Daptomycin and Bacitracin [56], Gramicidin S [57] and Bactenecin [58] most of these are extremely active and only some. Like Gramicidin S are allowed for topical Applications (wounds, burns, diabetic foot ulcers).
Cyclic Antimicrobial Cationic Peptides [59]
The antibacterial Agents that were triumphing in the 1960-1980 lose their effectivity and the pipeline where the more modern and with higher effectivity should be these days is practically empty. It is obvious that novel attitude must emerge, the efforts are spread, and people look for a bioactive agent which will eradicate the microbes, preferable to an unknown new mechanism. That is why back in the 1990s, the alarm was sounded regarding the collapse of antibiotic use due to resistance. Though the end was still decades away, the mere thought of l losing the ability to use these weapons for health defense sent a chill throughout the entire scientific community.
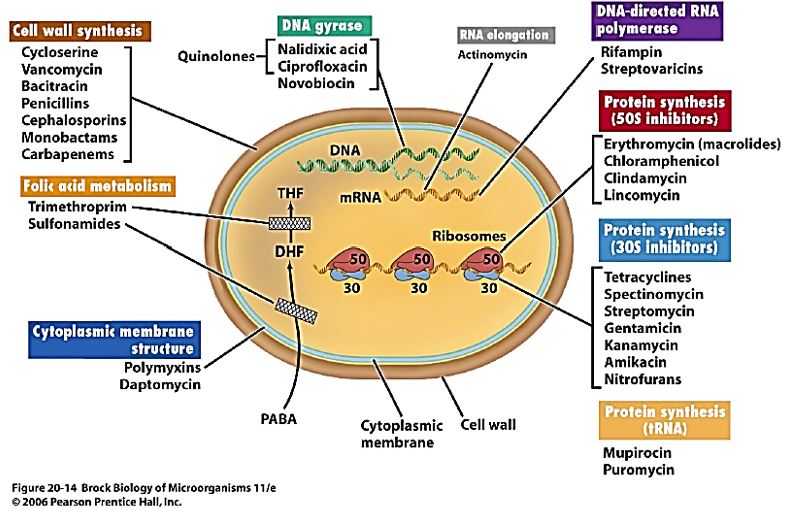
Figure 7 illustrates a part of the targets of various antibacterial agents. The art of drug production applied all of the modern technology from cybernetics to seek of active compound everywhere. In the deep jungles of Central America, Africa, and Asia, explored agent applied in the folklore medicine and by shamans, the result was stunning: a long list of agents that cured humankind of illnesses that were thought to be unmanageable like tuberculosis which is an infection based on mycobacteria.
The period 1960-1990 was a barren one concerning novel antibacterial drug discovery and development, only later, lipopeptides were developed as antibacterial drugs. Even today, the pipeline is practically dry, very few candidates exist.
Human illnesses such as pneumonia, diphtheria, scarlet fever, anthrax, and streptococcal and staphylococcal infections are caused by gram-positive bacteria - bacteria whose cell walls take up a violet stain known as Gram stain. In 1939, René J. Dubos (1901-1982) discovered gramicidin [60], an antibacterial agent that inhibits the growth of gram-positive bacteria. It was the first antibiotic to be tested clinically, and it was used in topical form to treat wounds and ulcers during World War II. More effective antibiotics soon superseded gramicidin. But Dubos’ discovery launched the antibiotic era and prompted other scientists to renew their stalled investigations into penicillin.
In the following tables one can find features of some main families of currently applied antibiotics [61].
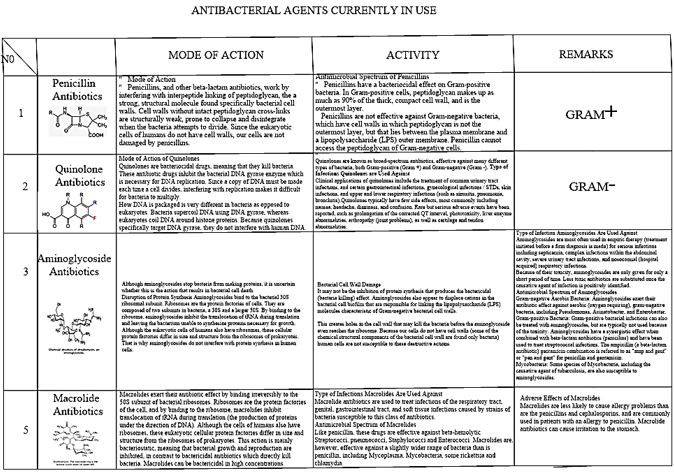
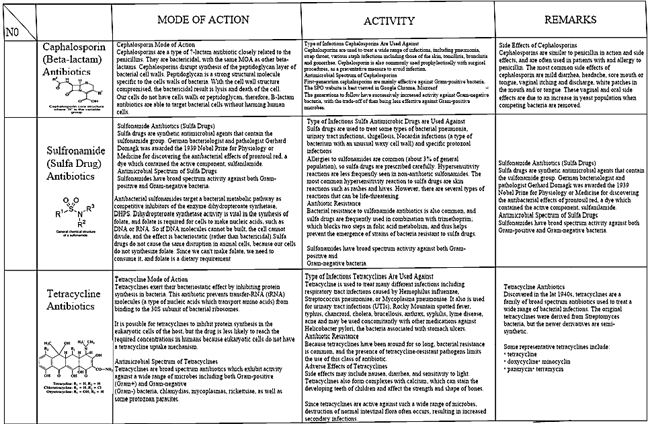
Medicine applied antibacterial agents abundantly. The list [62] of Antibiotic agents in a pharmacy is almost never-ending [63].
As part of the response, some researchers began the hunt for novel antimicrobial candidates in the environment. From soil to water to fish to bird to animals, any potential source was worth investigating. One of the most promising sources was reptiles [64]. Over the years, a handful of options were discovered from the skin of frogs, the venom of snakes, and even turtle eggs. Most of them took the form of antimicrobial peptides. These short chains of amino acids were found to have similar effects as antibiotics with little to no issues regarding resistance.
The discovery of Gramicidin S is considered to be the outcome of the intellectual transformation of Russian biologist G.F. Gause from simply a biologist to a researcher of antibiotics [65].
One of the encouraging results of these efforts is the focusing on the generally existing innate immune system of all creatures on earth, from plants through reptiles to humans: The “host defense peptide” or the “antimicrobial peptides” destined to combat microbes by nature.
Antimicrobial Peptides
The discovery of Gramicidine and its topical application for the cure of wounds in the early 1940 marks the
advent of the antibacterial agent’s era. However, one example is Magainin [66], which was discovered in
1987 by Prof. Michael Zasloff while he was doing genetic research on frogs. After several years of research,
Zasloff was surprised to discover that frogs that had undergone an operation recovered more quickly in the
non-sterile environment of an aquarium, without infection or inflammation. Zasloff hypothesized that a
frog’s skin secretes some sort of antimicrobial agent.
A few months later, he isolated the substance that was responsible for the frog’s quick recovery - a microbial peptide that he called Magainin (from the Hebrew word Magen– shield). Zasloff ’s research paved the way to identifying thousands of antimicrobial peptides in the plant and animal kingdoms, including mammals. Now, the era of antibiotics based on intracellular target components seem to end. Also, the era of antibiotics that hinder cell wall synthesis is losing momentum. The long list of antibiotic drugs to its families is becoming insufficient since it hardly answers the common bacterial infections acquired nonsocial or otherwise. It seems that an exploitation of the Antimicrobial Peptides (AMP, Examples include Cecropins, from insects, Magainins from amphibians and Cathelicidins, from mammals) and their way to eradicate bacteria by cell wall disruption is one of the few clues to the problem of microbe’s resistance, eradication of persister cells, dormant cells and mutants may become a real option and bring remedy to those who suffer the incurable infection caused by those killer bacteria [67].
The antimicrobial peptides are nature’s weapon to combat the invasion of microbes as host defense agents, the first line of defense. However, the natural peptides suffer from serious drawbacks the first in the list is their being peptides and sensitive to enzymatic digestion.
Peptides are recognized for being highly selective and efficacious and, at the same time, relatively safe and well tolerated. Consequently, there is an increased interest in peptides in pharmaceutical research and development (R&D), and approximately 140 peptide therapeutics are currently being evaluated in clinical trials. Given that the low-hanging fruits in the form of obvious peptide targets have already been picked, it has now become necessary to explore new routes beyond traditional peptide design. Here is a pro- and contra- reasons for the use of antimicrobial peptides as active drugs [68]:
Development [69] of Antimicrobial Peptides [70] as Anti-Infective Drugs [71]
Advantages
Broad-spectrum activity (antibacterial, antiviral, antifungal)
Rapid onset of killing
Cidal activity
Potentially low levels of induced resistance
Concomitant broad anti-inflammatory activities
Disadvantages
Discovery costs of synthesis and screening
Patent exclusivity for economic viability
Systemic and local toxicity
Reduced activity based on salt, serum, and pH sensitivity
Susceptibility to proteolysis
Pharmacokinetic (PK) and pharmacodynamic (PD) issues
Sensitization and allergy after repeated application
Natural resistance (e.g., Serratia marcescens)
Confounding biological functions (e.g., angiogenesis)
High manufacturing costs
In 2006 (currently 140 are in the pipeline), only one anti-infective with the topical application [72] has shown efficacy in Phase III clinical studies (omiganan pentahydrochloride 1% gel; Migenix), a bovine indolicidin-based peptide, was developed for the prevention of contamination of central venous catheters. In a completed Phase III study, MX-226 demonstrated a statistically significant 49% reduction of local catheter site infections, as well as a 21% reduction of catheter colonization. Cadence is currently conducting a confirmatory. Phase IIIb study of MX-226 for the prevention of local catheter site infections. Other current trials involve completed Phase II clinical trials against mild-to-moderate acne (indolicidin-based MX594AN; Migenix) and completed Phase I trials for the prevention of infections in patients undergoing allogeneic stem cell transplantation (human lactoferricin-based hLF1-11; AM-Pharma [http://www.ampharma. com]). Antimicrobial peptides are currently used clinically in two topical and two systemically applied formulations for the treatment of several diseases, as well as prophylactically to prevent infections in neutropenic or cystic fibrosis patients. Topical applications of polymyxins (polymyxin B and polymyxin E) and gramicidin S in the treatment of infections caused by P. aeruginosa and Acinetobacter baumannii are clinically safe and effective, with little development of resistance. Polymyxin B is a cyclic 10-amino-acid cationic antimicrobial lipopeptide that also binds and neutralizes endotoxin. Unfortunately, both polymyxins and gramicidin S are too toxic at clinically used doses to be utilized systemically as anti-bactericidal or antiendotoxic reagents. A topical combination of polymyxin B and gramicidin S (or neomycin) are routinely used clinically, often together with bacitracin for generic wound creams, eye drops and tear drops.
The approach of peptidomimetics (Figure below) [1] bears many advantages over the gift of nature [73],
and that is:
1. The stability, in the enzymatic world.
2. The option of human design.
3. Use of un-natural components to the construction of the mimetic architecture.
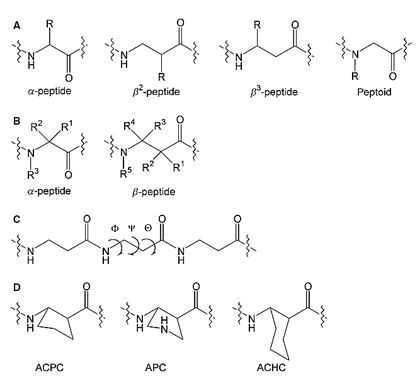
An approach based on those lines enables the introduction of the novel design of components that may aid in the eradication but are novel for nature. It may allow the exploration of the way of selective eradication of microbes in the presence of beneficial flora and point on the cure of cancer [74].
There are many obstacles in this way: It is needed to learn about the mechanisms in which the AMP disrupt the cell membranes in full detail. Today, many ways were found in which the membranes are destroyed. It was found that low molecular weight (MW = 500) non-peptide mimics can eradicate bacteria, and small structural modification may lead to preference in eradication at the will of either Gram-positive or Gramnegative bacteria.
Despite several attempts to develop AMPs as antibiotics [75], the reasons why synthetic AMPs have not progressed more successfully through the clinic include the cost of goods, their lability to proteolytic degradation, and their unknown toxicology profile when administered systemically. Each of these factors can be addressed by the peptide design approaches described above in combination with advanced cheminformatics tools.
For example, the cost of goods can be addressed by making smaller peptides, and machine-learning approaches have already delivered highly active, broad-spectrum peptides that work systemically in animals. The liability to degradation by proteases in the body can be addressed using d amino acids, non-natural amino-acid analogues, mimetics with different backbone structures or appropriate formulations.
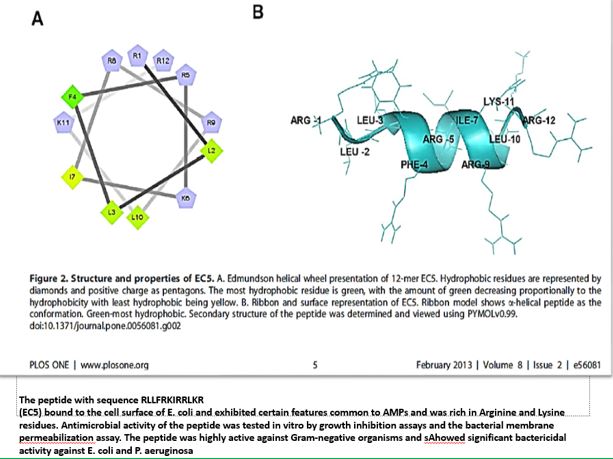
The Challenge
Resistance to antibiotics has increased dramatically over the past few years and has now reached a level that
places future patients in real danger. Microorganisms such as Escherichia coli and Klebsiella pneumoniae,
which are commensals and pathogens for humans and animals, have become increasingly resistant to thirdgeneration
cephalosporins [46]. The interactions of an AMP with the membrane cannot be explained
by a particular sequential amino-acid pattern or motif; rather, they originate from a combination of
physicochemical and structural features including size, residue composition, overall charge, secondary
structure, hydrophobicity and amphiphilic character.
Pore formation by interaction with cell wall lipids and the changes in permeability and with it the ease of penetration of the AMPs through the forming pores is determining the effect on the eradication difference which is a result of disruption of the plasma membrane of the bacteria. Moreover, interactions with the many components that furnish the architecture of the membranes are crucial for antibacterial activity.
There are drawbacks to the use of peptides as therapeutics. The low bioavailability and high cost. These obstacles may be overcome since a great deal of effort is being conducted to circumvent the problems associated with various methods including the use of D- or unnatural amino acid, formulation, recombinant DNA expression of peptides, the addition of fatty acyl chains to short peptides. Therefore, it is expected that antimicrobial peptides will become the drugs of choice for emerging bacterial infections in the future [76].
The approach of design and synthesis of mimics of active motifs of the antimicrobial peptide, their artificially prepared surrogates, presents wider scope and with it a reasonable outlet of the un-favored situation in which natural AMP is at present, considered as an antimicrobial agent for the combat with the nosocomial microbe’s pandemic. Many of the drawbacks of the AMPs are to be overhauled by this approach since the surrogates are in their nature nonpeptide and allow the introduction of unnatural elements into their molecular structures.
The Research and Discovery of Drug-Like Molecules
The systematic research work begins with the isolation and identification of the chemical structure of the
natural peptide. Until today, approximately 2000 AMP have been identified in this way from creatures
and plants of all kingdoms of living organisms on earth. The structures and sizes (from 5-50 amino acids
sequences) are presenting a variety of structure, but most of them are amphipathic, cationic peptides with
characteristics of hydrophobic and hydrophilic zones in the polypeptides. Many are based on the cationic
amino acids Lysine and Arginine.
The next step is the identification of reactive motif by way of preparing/isolating linear epitopes. The general research endeavor can be summarized as follows in the scheme 1[77a,77b]:
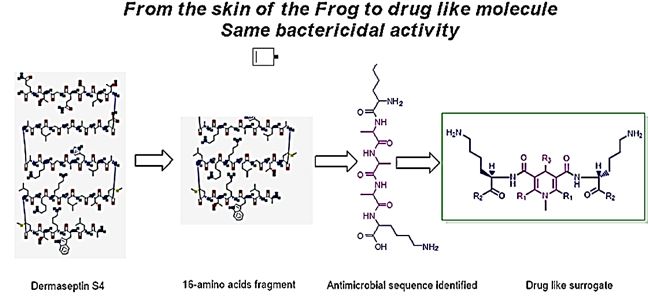
Scheme 1: The identification of active sequence and mimic as “Drug Like Surrogate.”
The guidelines in the surrogate design are:
1. Retain the amphipathic nature of the AMP in the surrogate
2. Retain the charges present in the active motif
3. Introduce new (not present) in the AMP like β-turn mimics [78].
4. Keep the biological activity (eradication of bacteria, no hemolysis of erythrocytes) of the surrogate in the
ranges of the motif.
5. Keep the Lipinski rule of 5 in the surrogate [79].
Despite several attempts to develop AMPs as antibiotics [80], the reasons why synthetic AMPs have not progressed more successfully through the clinic include the cost of goods, their lability to proteolytic degradation, and their unknown toxicology profile when administered systemically. Each of these factors can be addressed by the peptide design approaches described above in combination with advanced cheminformatics tools. For example, the cost of goods can be addressed by making smaller peptides, and machine-learning approaches have already delivered highly active, broad-spectrum peptides that work systemically in animals. The liability to degradation by proteases in the body can be addressed using d-amino acids, non-natural amino-acid analogues, mimetics with different backbone structures or appropriate formulations.

Synge [81] explored the cyclic AMP Gramicidin S [82], which is applied today for topical antibacterial therapy. Katchalski (Katzir) prepared poly-lysines and explored their biocidal quality [83]. Pexiganan is a mimic of Magainin. Its surrogate Pexiganan, (or LocilexTM), as the therapeutic cream was named. It was specific for bacterial membranes (mammalian membranes do not carry an external negative charge), attacked a large spectrum of bacteria, and bacteria would not easily develop resistance to it because it does not bind to a discrete and mutable bacterial target [84]. LTX 109 is an experimental drug which was developed in Norway as a Synthetic Antimicrobial Peptidomimetic (LTX 109) along the lines indicated above [85]. It is meant to aid in the cure of diabetic ulcers, for topical application only.
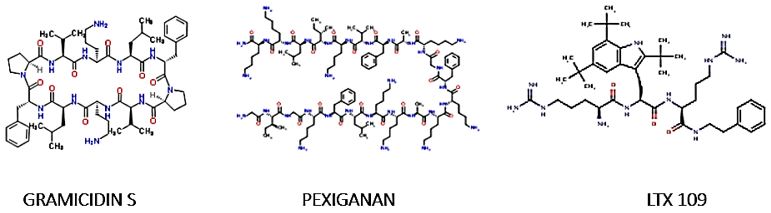
Literature survey reveals that about 6 efforts are operating these days aiming at surrogates of active antimicrobial peptide fragments (motifs) as follows:
The general strategy is applied by many to the crucial turning point where such motifs are identified [86]. Then the mimics designed, synthesize and check for antibacterial activity and toxicity. A usual procedure uses MIC determination an human erythrocytes hemolysis.
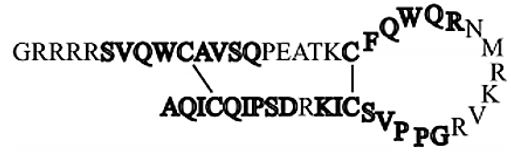
An example is the identification of an antibacterial motif in Lactoferricin [87] as follows. Active fragments were translated to a drug candidate by the company LTX 109 (above):
Now, one can summarize some of such antibacterial as in the next chart:
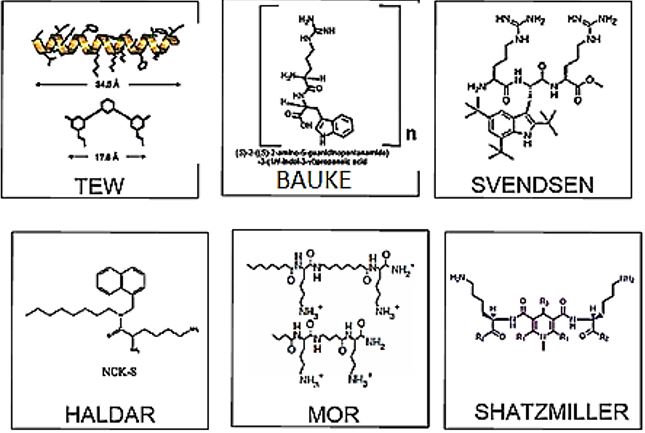
Tew has taken this approach to the point where he expressed the idea in showing that the amphipathic motif of Magainin 2 [88] is in fact translated to a hydrocarbon scaffold, carrying the tow β- amino groups providing a hydrophobic scaffold with two flanking positively charged amino units.
The effort in this frame was to identify short bioactive (antimicrobial) peptide sequences and in AMP from various sources (Human, Amphibians, Fish, insects, Serial, etc. In the following drawing are represented a few structure of surrogate antimicrobial structure that was designed by natural motifs found in AMPs.
Tew [89] and his colleagues designed the tricyclic aromatic structure by the general idea of amphipathic nature of AMP, in particular, the work of Vogel [90]. Park [91] and Bauke [92] explored the motifs WK and WR respectively as a unit for the eradication of bacteria. Svendsen ans partners established a corporation for the development of antibacterial medicins based on Lactoferricin turn [93,94]. On this basis, the Topical drug molecule LTX 109 was developed by Svendsen and the group from LYTX [95]. Haldar and his group investigated the use of [96] Aryl-Alkyl-Lysines as Agents That Kill Planktonic Cells, Persister Cells, Biofilms of MRSA and Protect Mice from Skin-Infection. Mor and his coworkers applied an amphipathic lysine unit to construct antimicrobial polymers [97].
Lantibiotics
The name lantibiotics, derived from a class of antimicrobial peptides that contain a thin ether bridged
dipeptide moiety, a lanthionine, while in general lanthionine - containing peptides are called lanthipeptides.
The Lantibiotics, divided into two different classes according to their structural features. The lantibiotic
family is subdivided into two groups - the type I and II lantibiotics - that comprise peptides with straightchain
and globular structures, respectively. Most, if not all, lantibiotics target components of the plasma
membrane of bacteria, with more than one-third of the known lantibiotics targeting Lipid II.
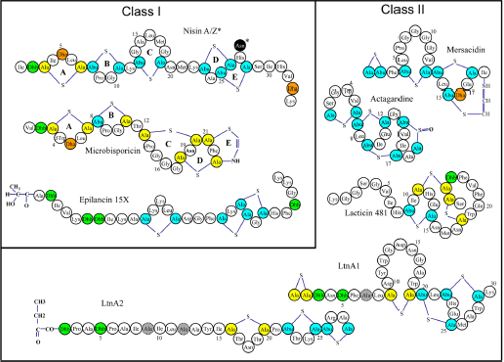
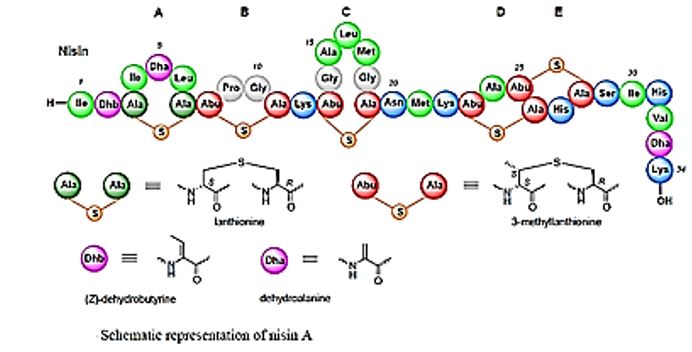
Type-I lantibiotics are cationic, elongated peptides up to 34 residues long, which act on the bacterial cell membrane, with the lantibiotic nisin as its most known representative example, as shown in Figure (above). Type II lantibiotics have a more globular structure, up to 19 residues in length, and act through disruption of enzymatic functions [98].
Only a few naturally occurring antimicrobials, such as the mostly applied lantobiotic agent from natural source nisin, the macrolide -glycoside natamycin, and the protein lactoferrin and the enzyme lysozyme, have regulatory approval for application to foods. Many additional antimicrobials, especially those derived from microorganisms, hold the potential for regulatory approval in the future [99].
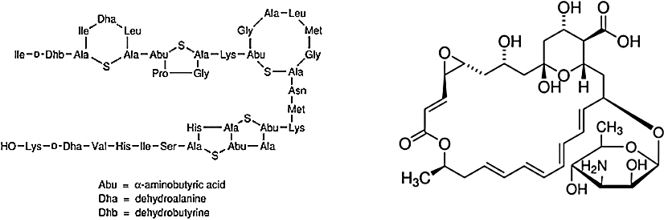
The lantibiotics continue to attract the most attention. The implementation of in vivo and in vitro engineering systems to alter, and even enhance, the antimicrobial activity, antibacterial spectrum and physicochemical properties, including heat stability, solubility, diffusion and protease resistance, of these compounds. Additionally, we discuss the potential applications of these lantibiotics for use as therapeutics [100]. Lantibiotics possess many of the attributes essential for the treatment of infections caused by multidrug resistant bacteria and their potential for use as alternatives to traditional antibiotic therapies has been mooted for decades.
The antibacterial activity of niacin relays on a complex with lipid II which is an important cell wall precursor component in bacteria essential for cell wall construction. It is peptidoglycan, which is amphipathic and named for its bactoprenol hydrocarbon chain, which acts as a lipid anchor, embedding itself in the bacterial cell membrane. Lipid II must translocate across the cell membrane to deliver and incorporate its disaccharidepentapeptide “building block” into the peptidoglycan mesh. Lipid II is the target of several antibiotics: Lipid II as a target for antibiotics [101].
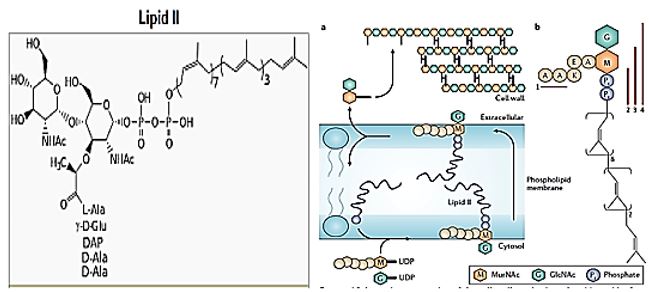
Schematic presentation of the cell-wall synthesis cycle. A | Assembly of the cell-wall subunit takes place on the cytosolic side of the bacterial plasma membrane. UDP-activated precursor sugars are assembled on a polyisoprenoid carrier, the coupling of which produces Lipid II, which is then transported across the membrane by an unknown mechanism. Next, the peptidoglycan subunit is transferred to the growing peptidoglycan chain and the polyisoprenoid carrier is recycled back to the cytoplasmic side to complete the cycle. B | Structure of Lipid II. The polyisoprenoid anchor consists of eight isoprene units in the cisconformation followed by two units in the trans-conformation and the terminal isoprene unit. Depicted here is a Lipid II carrying a pentapeptide with lysine at the third position.
Together with diaminopimelic acid, this is the most common residue at this position. The third amino acid of the pentapeptide is coupled to the glutamate at position two via the side chain carboxylate. The red bars indicate the minimal binding sites in Lipid II of glycopeptide antibiotics (1), nisin (2), ramoplanin (3) and mersacidin (4). GlcNAc, N-acetylglucosamine; MurNAc, N-acetylmuramic acid.
Niacin was used to eradicate Gram Positive 3 bacteria in microorganisms colonies to enable the collection of Gram Negative bacteria, fungi and other microbes. Now, their are 3 report on the the 3 use of niacin in infection caused by Gram-Negative (salmonella) bacteria [102].
Late 1930 were stormy also in antibacterial research. Gramicidin S attracted the attention of many scientists including that of the Nobel Laureate Synge [103]. The discovery of the high antibacterial activity stimulated many researchers, including Katchalski [104] (Kazmir) and his group to pursue research in the new area of polymeric amino acids. In another publication, focusing on α- polylysine, Katchalski and colleagues report on the broad spectrum antibacterial activity [105]. However, it was also established that those polymeric agents are toxic to the human blood tissue, The cause agglomeration of the blood [106]. Polylysine attracted the attention of many [107], however, ε-polylysine was also found to be antibacterial [108]. The antibacterial application of Polylysine for protection of surfaces of metals have been reported [109].
In recent years, a n enormous wave of activity is discovering novel forms and application of these antimicrobial polymers as antibacterial agents. Kenichi Kuroda and Gregory A. Caputo reported on antimicrobial polymers as synthetic mimics of host - defense peptides [110]. Antimicrobial-peptide mimetic polymers display antibacterial activity against a broad spectrum of bacteria including drug-resistant strains and are less susceptible to resistance development in bacteria. These polymers also showed selective activity to bacteria over mammalian cells. Antimicrobial polymers provide a new molecular framework for chemical modification and adaptation to tune their biological functions. The peptide-mimetic design of antimicrobial polymers will be versatile, generating a new generation of antibiotics toward implementation of polymers in biomedical applications.
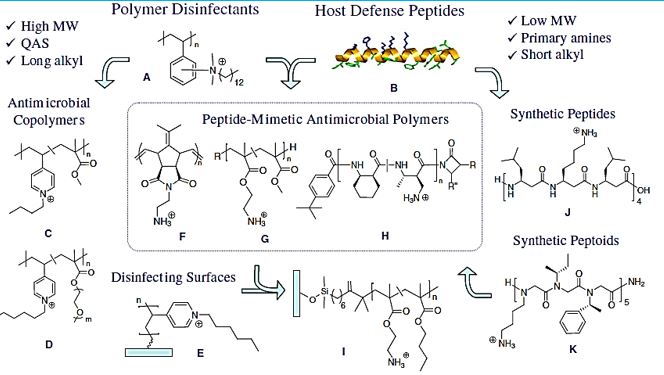
Edmund F. Palermo & Kenichi Kuroda investigated antibacterial polymers on surfaces [111]. Based on the idea the activity of the polymers can be modulated by tuning key structural parameters, they outline the design principles that have been developed so far to fine-tune the activity of these antimicrobial agents.
Degrado and colleagues presented an account of the activity in this area [112]. In their publication, the researchers try to mimic the features of the antimicrobial polypeptides, based on artificial surrogated of the building blocks. “Because the activities of many AMPs depend primarily on their overall physicochemical properties rather than the fine details of their precise amino acid sequences, we have designed and synthesized very small “coarse-grained” molecules, which are far simpler than naturally produced AMPs. The molecular design of these foldamers epitomizes the positively charged amphiphilic structures believed to be responsible for the activity of AMPs. The designed oligomers show greater activity than the parent peptides”.
Mor and collaborators investigated the lysine-based oligomers as mimics of antibacterial peptides [113]. The group prepared oligomers of acylated lysines (OAKs) are synthetic mimics of host defense peptides (HDPs) with promising antimicrobial properties.
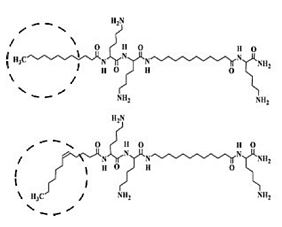
The resulting compound, (molecular structure is shown in Fig. above), displayed reduced self-assembly and hemolysis but maintained potent, albeit selective, antibacterial activity.
In the focus: the amino acids tryptophan, valine, Arginine and Lysine.
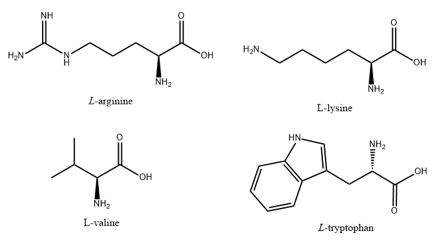
The balance between charge and lipophilicity in the antimicrobial peptides (or their surrogates) might be a major factor in their effectivity as antibacterial agents. Agents that in connection to The interactions of an AMP with the membrane cannot be explained by a particular sequential amino-acid pattern or motif; rather, they originate from a combination of physicochemical and structural features including size, residue composition, overall charge, secondary structure, hydrophobicity and amphiphilic character. Pore formation by interaction with cell wall lipids and the changes in permeability and with it the ease of penetration of the AMPs through the forming pores is determining the effect on the eradication difference which is a result of disruption of the plasma membrane of the bacteria. Moreover, interactions with the many components that furnish the architecture of the membranes are crucial for antibacterial activity. From our study, we conclude that the venerability of bacteria may depend on small structural variation in the composition of the biocide.
Those four amino acids were identified as great contributors to antibacterial activity in antibacterial peptides.
Chinese research indicates that there are strand length-dependent antimicrobial activity and membraneactive mechanism of arginine- and valine-rich-hairpin-like antimicrobial peptides [114], Cutrona, Kaufman, Figueroa, and Elmore report on the Role of Arginine and Lysine in the Antimicrobial Mechanism of Histone-derived Antimicrobial Peptides [115]. Motifs containing lysine–tryptophan motifs [116] are also reported in the same effort to extract the essential combination of amino acids in antibacterial motifs that may show the way in the design process. Specifically, lysine (K) or arginine (R)-tryptophan (W or Trp) or isoleucine (I)-phenylalanine (F) motifs have been designed for bacterial membrane selectivity [117,118]. However, KW, RIF or RW series peptides show increased hemolytic activity and hydrophobicity with higher chain length [116,117]. In a separate study using a de novo designed alpha helical HDP, high hydrophobicity was found to correlate with strong hemolytic activity [119].
Many mechanisms are in discussion for the eradication of bacteria withy AMPs [120]. We can take the attack of a small peptide isolated from honeybees [121]. Apidaecin on bacteria cells as
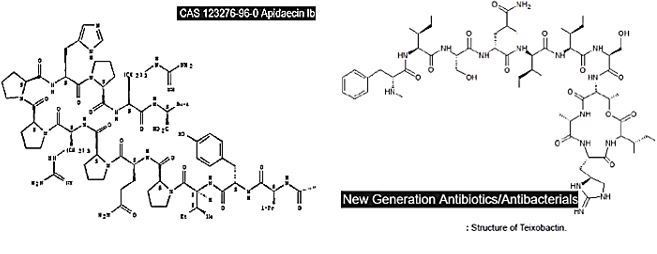
a working hypothesis: The mechanism of action by which Apidaecins kill bacteria involves an initial “surface state” non-specific binding of the peptides to an outer membrane component. This binding is followed by the invasion of the periplasmic space and by a specific and essentially irreversible combination with a receptor/docking molecule that may be a component of a permease-type permeability and transport system on inner membrane. In the final step, the peptide is translocated into the interior of the bacterium where meets its ultimate target [122].
Recently, evidence was if killing takes place only when bacterial cell membranes are completely saturated with AMPs. This condition is achieved for all bacteria. However, Since the in Gram-negative bacteria the outer membrane are crowded, packed with various proteins (up to 50% of the total membrane weight [123]), compared of only 15% in the S-layer (surface layer) [124] in gram-positive. This difference demands more energy for saturation in gram-negative than in gram-positive bacteria. The Gram-negative bacteria are surrounded by two membranes both have to be disassembled for eradication, whereas only one membrane protects the Gram-positives.
In this situation, where the mechanism of action of the antimicrobial peptide is unclear, it id very cumbersomely to initiate a major effort aiming at the eradication of bacteria applying drugs in a known manner. The efforts of Norwegian scientist regarding the mimicking of lactoferricin part structure may lead to an applicable drug material.
John-Sigurd Svendsen and his collaborators investigated the peptide sequences by preparing synthetic cationic antimicrobial peptides (CAPs) studied in this work have been developed based on truncation and systematic mutations of lactoferricin [12], a natural AMP found in milk present in milk [125-127].
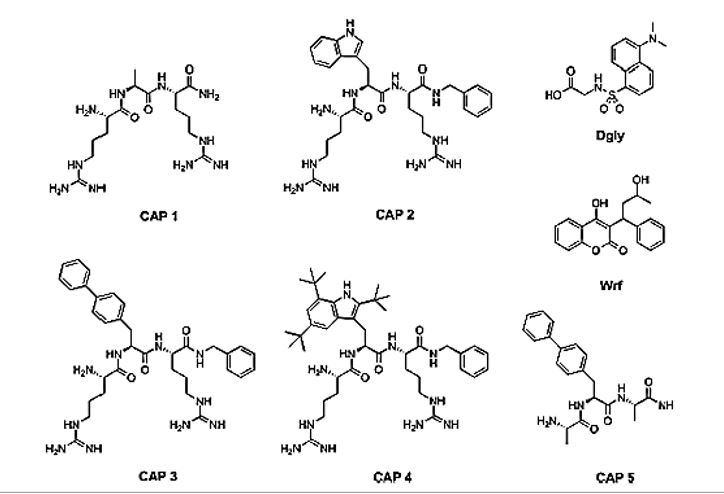
Non-Peptide Antibacterial Agents
The antimicrobial peptides with the various modification and diversity, are the agents produced and
applied by all living beings on earth as a tool applied in the innate immune system to combat microbes by
eradication. However, there are many families of natural products that can eradicate microbes. In this part of
the composition, we would like to survey this field. Other compounds types have been identified as potential
antibacterial agents: Acetylenes, Coumarins, Flavonoids and isoflavonoids, Iridoids, Lignans, Macrolides,
Phenolics (other than flavonoids and lignans), Quinones, Steroidal saponins, Terpenoids, Xanthones, and
more [128].
Did anybody ask the question “In a world without antibiotics, how did people treat infections? “ Well, traditional medicine in Africa, for example, applies herbs and plants in case of infection to cure [129]. However strange as it may seem, the plants contain many antibacterial compounds in Herbs and spices, fruits and vegetables, seeds and leaves, and other parts of the vegetation organisms as follows:
Plants produce an array of secondary metabolites that can be found in the edible, medicinal, and herbal plants and their derived essential oils (EOs) [130]. Secondary metabolites from plants are extensively studied as promising healthy ingredient or human disease controlling agents. These secondary metabolites possess various benefits including antimicrobial properties against pathogenic and spoilage microbes [131]. Natural antimicrobials derived from plants have been recognized for centuries, but only scientifically confirmed in the last 30 years [132,133]. Thus an increasing interest in finding natural antimicrobials for application in food products to prevent or inhibit microbial growth and extend shelf life has been noticed [134,135]. As one example, Substances from bergamot peel, a byproduct of citrus fruit processing, was found to be active against Gram-negative bacteria (Escherichia coli, Pseudomonas putida, Salmonella enterica) and the antimicrobial potency of flavonoids was increased after enzymatic deglycosylation [136]. Natural products are both a fundamental source of new chemical diversity and an integral component of today’s pharmaceutical compendium [137]. However, many currently available antifungal and antibacterial agents have undesirable toxicity, and the widespread use of these drugs has led to rapid development of drug-resistant strains, which are the leading cause of failure in both clinical and agricultural applications.
Of particular antibacterial agents are Alkaloids, Acetylenes, Coumarins, Flavonoids and isoflavonoids, Iridoids, Lignans, Macrolides, Terpenoids and Phenolics (other than flavonoids and lignans). Examples are in the following drawings:
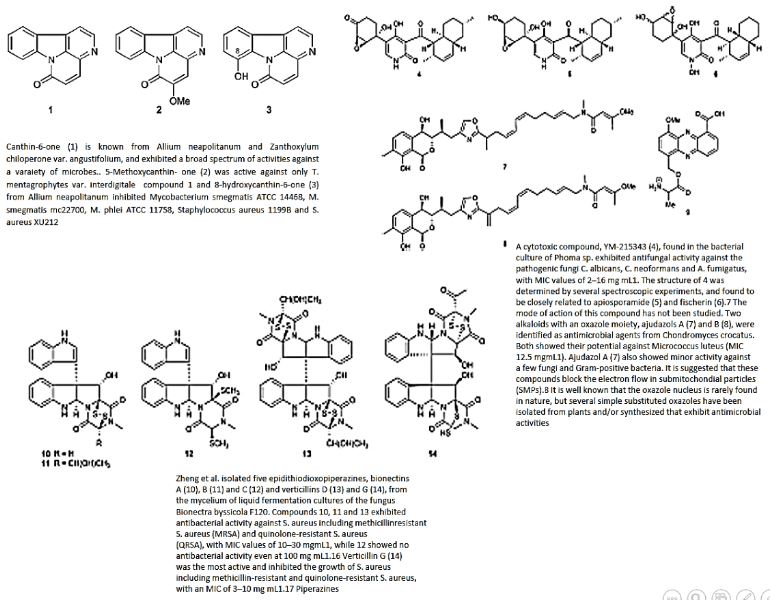
Alkaloids
The alkaloids from different organisms (plant, bacteria, fungi) and their biological activity in the eradication
of microbes are in the following chart
Acetylenes
The acetylenic antibacterial compounds, Compared to saturated fatty acids, unsaturated fatty acids with
double and/or triple bonds are, in general, more potent against fungal pathogens [138].
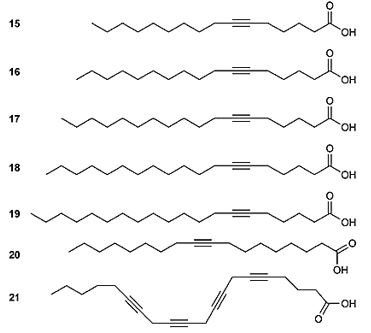
Five 6-acetylenic acids (15-19) purified by reverse-phase HPLC were identified as potential antimicrobial chemicals, their structures being established by LC-MS, NMR, and HPLC-ESI-MS studies. Compounds 15-18 showed antibacterial activity, while compound 19 was not active. Reference comp most active, in particular against the dermatophytes Trichophyton mentagrophytes and T. Tak potencies, these 6-acetylenic acids (15-21) may be excellent leads for further preclinical studies [139]. The macrolides are amonmg thye terpenoids of extreme therapeutic significance.
Macrolides
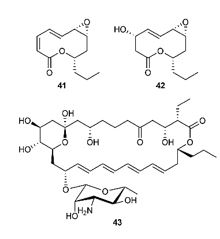
Macrolides are known to be bacterial constituents with significant antimicrobial activities. Several macrolides have been identified and evaluated for their activities [140], and in recent years work has been in progress to isolate such potential antimicrobials, mostly from microbial sources. Two 10-membered macrolides, phonolites A [141]and B [142], were purified from the culture of Phomopsis sp. hzla01-1. Both compounds showed significant antimicrobial activities against Escherichia coli, Candida albicans and Saccharomyces cerevisiae, with MICs in the range of 5-10mg mL1. Furthermore, these compounds showed macrolide, a secondary metabolite of Streptomyces diastaticus, also exhibited potent antimicrobial properties against Aspergillus candids, Fusarium oxysporum, Aspergillus niger, Microscporum gypseum and Trichophyton Microsporum canis, Trichophyton rubrum and Trichophyton tonsurans (MICs 16mg mL1). This compound should therefore be a good candidate to develop a broad-spectrum antifungal drug [143].
Terpenoids
Terpenoids are a class of secondary metabolites made of isoprene units. The important phytochemicals in
essential oils are mixtures of mono- and sesquiterpenoids, and are known for their antimicrobial properties
- 60% of essential oil derivatives examined to date are inhibitory to fungi, while 30% inhibit bacteria [144].
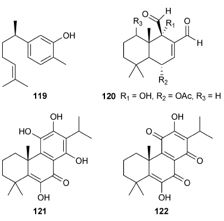
These facts indicate the antimicrobial potential of this important class of compounds. The sesquiterpene xanthorrhizol (119), isolated from the ethanol extract of Curcuma xanthorrhiza, has antibacterial activity. In addition, it maintained its antibacterial activity after thermal treatment a wide range of pH conditions (pH 3.0, 7.0 and 11.0), strongly suggesting that it could be effective as a natural preservative to prevent the growth of foodborne pathogens. Cinnamodial (120), a diterpenoid isolated from the leaves and bark of Pleodendron costaricense, exhibited high activity against Alternaria alternata (MIC 3.9mg mL_1), and other microbes. Wellsow et al. investigated Plectranthus saccatus for itsantimicrobial secondary metabolites, and isolated coleon U (121) and coleon U quinone (122). Compound 121 showed high activity against B. Subtilis and Pseudomonas syringae, with an MIC of 3.13 and 6.25mg mL, respectively, and 122 also hadhigh activity against P. syringae, with an MIC of 3.13mg mL.
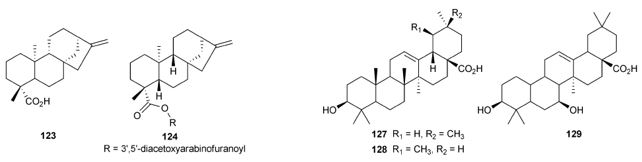
Pentacyclic triterpenes also have antimicrobial activities,many of them being found in various plant families.Oleanolic acid (127) and ursolic acid (128) are well-known phytochemicals, isolated by Horiuchi et al. from the extract of the leaves of Salvia officinalis Both 126 and 127 exhibited potent activity against vancomycinresistant Enterococcus (Streptococcus pneumoniae and MRSA, with MICs of 16.0 and 8.0 mg mL_1, respectively [145].
Previously, Woldemichael et al. and Kowalewski et al. had described the antimicrobial properties of (127) and (128) [146a,b], with quite different results. It seems that such differences are due to differences in assay conditions and in the strains used. Both (127) and (128) have very low toxicity - 127 has already been successfully used as an orally administered drug to treat human liver diseases in China, 104 and so they might be used for the treatment of VRE infections. Compound 127, together with another pentacyclic triterpenoid, canthic acid (129), was isolated from the root bark of Newbouldia laevis. Both compounds showed broad-spectrum in vitro antimicrobial activity against six Grampositive and twelve Gram-negative bacterial species, as well as Candida strains (MICs 0.038-9.76mg mL_1); notably, 129 showed activity of 0.038mg mL_1 against Bacillus Subtilis and B. Cerus and 0.076mg mL_1 against B. Megaterium [147].
Miscellaneous Compounds
The cytotoxic compound aculeatin D (141) was isolated as minor constituent from the rhizomes of Amomum
aculeatum, and showed very potent activity against two Plasmodium falciparum strains [148]. The cytotoxic
compound aculeatin D (141) was isolated as minor constituent from the rhizomes of Amomum aculeatum,
and showed very potent activity against two Plasmodium falciparum strains Platensimycin [149] (142) is a
novel broad-spectrum Gram-positive antibiotic produced by Streptomyces platensis, and was discovered by
a target-based whole-cell screening strategy using an antisense differential sensitivity assay.
The cytotoxic compound aculeatin D (141) was isolated as minor constituent from the rhizomes of Amomum aculeatum, and showed very potent activity against two Plasmodium falciparum strains Platensimycin (142) is a novel broad-spectrum Gram-positive antibiotic produced by Streptomyces platensis, and was discovered by Mechanistically, it exerts its activity by a novel mode of action that involves specific binding with the acyl enzyme intermediate of the key condensing enzyme FabF of the fatty acid synthesis pathway. Fatty acids are essential for survival of bacteria and are synthesized by a series of enzymes including the elongation enzymes, b-ketoacyl acyl carrier protein synthase I/II (FabF/B). Inhibition of fatty acid synthesis is therefore a target for the discovery and development of antibacterial agents, and 142 could be a candidate for antibacterial drug development. [150]
Nonpeptide Antibacterial Polymers
Microbial infection remains one of the most serious complications in several areas, particularly in medical
devices, drugs, health care and hygienic applications, water purification systems, hospital and dental surgery
equipment, textiles, food packaging, and food storage. Antimicrobials gain interest from both academic
research and industry due to their potential to provide quality and safety benefits to many materials.
However, low molecular weight antimicrobial agents suffer from many disadvantages, such as toxicity to the
environment and short-term antimicrobial ability. To overcome problems associated with the low molecular
weight antimicrobial agents, antimicrobial functional groups can be introduced into polymer molecules. The
use of antimicrobial polymersoffers promise for enhancing the efficacy of some existing antimicrobial agents
and minimizing the environmental problems accompanying conventional antimicrobial agents by reducing
the residual toxicity of the agents, increasing their efficiency and selectivity [151]. The case of using these
antimicrobial agents in food packaging, there is a risk of diffusion of these agents into the food causing
various problems [152]. In water treatment, the most popular treatment method to disinfect and sterilize
water is to use chlorine and other related chemicals. However, their residues can become concentrated in
the food chain and in the environment as well as the possible formation of halomethane analogues that
are suspected of being carcinogenic should lead to the avoidance of their use [24]. Due to the associated
problems result from the use of conventional antimicrobial agents; the idea of polymeric antimicrobial agents
appeared to be an attractive alternative. Nowadays, there is an increasing interest in selective antimicrobial
polymers whose potency against bacteria and non-toxicity towards mammalian cells distinguishes them
from most polymeric biocides that are broadly poisonous [153a-153g].
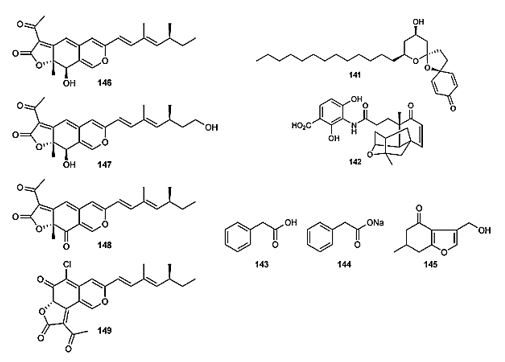
It is well known that the bacterial cell surface is usually negatively charged as evidenced by its susceptibility to electrophoresis. Adsorption of polycations onto the negatively charged colloidal surface is expected to take place than that of monomeric cations. Therefore, it is reasonable to assume that in the elementary process step (1), figure 4, is enhanced for polymers as compared to that for monomers. Ikeda and co-workers synthesized polycationic biocides with pendent phosphonium salts and compared their antibacterial activity with the corresponding monomers (Figure 2). They also demonstrated that the activity is increased in the order of increasing molecular weight. Chen and co-workers synthesized quaternary ammonium functionalized\ poly (propylene amine) dendrimers (Figure 4) [154a-154f]. They found that the antimicrobial properties of these dendrimer biocides have parabolic dependence on molecular weight [155]. Tokura and co-workers found similar results [156].
regard to their interactions with bacteria is quantitatively summarized in Table 1.37 However, Panarin and co-workers reported that the bacteriostatic properties had no molecular weight with pendent quaternary ammonium groups [157]. 39 To explain properly the discrepancy of the trends in these data, one needs to take into consideration the bacteria structure. Depending on the sophistication of the cell wall structure, bacteria can be divided into two classes, Gram-positive (e.g., S. aureus) and Gram-negative (e.g., E. coli). Gram-positive bacteria tend to have a loose cell wall, while Gram-negative bacteria have an outer membrane structure in the cell wall forming an additional barrier for foreign molecules. Most of the investigations that were summarized above deal with the molecular weight dependence on S. aureus eradication. Studies indicated that molecules with molecular weight up to 5 _ 104 to 9 _ 104 Da do not seem to have problems diffusing across the cell wall of the Gram-positive Bacterium S. aureus. For Gram-negative bacteria, such as E. coli, the question of diffusion to the cell membrane is even more complicated due to the existence of an outer membrane.
Effect of Counterion.
Kanazawa and co-workers investigated the counter anion dependence of poly
[tributyl(4- vinylbenzyl)phosphonium] salts (Figure 3) against S. aureus. The antibacterial activity was found
to be affected by the structure of the counter anions.20 The activity was low for a counteranion, which tends
to form a tight ion-pair with phosphonium ion, while the order of chloride > tetraflouride > perchlorate >
hexafluoro phosphate, which could be correlated with the solubility products of the polymers. The antimicr
the counterion. They found that the biocides with bromide anions are more potent than those with chloride
anions.37 However, the work carried out by Panarin and co-workers on the synthesis of photopolymers of
vin counter anions on the antibacterial activities among chloride, bromide, and iodide.39 It is not clear why
counterions should have an effect on antimicrobial activity except where they alter solubility of their host
polymers.
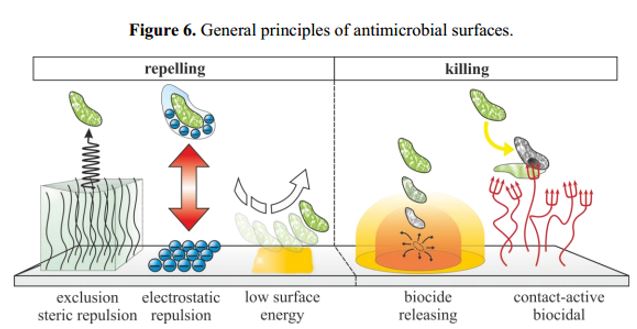
Antibacterial Coating of Surfaces
How do the bactericides work from the surface [158]? Biofilms on materials are extremely hard to remove
and show great resistance to all kinds of biocides. Thus, the prevention of biofilm formation by antimicrobial
surfaces is the best way to avoid spreading of diseases and material deterioration. To do this, the material
must avoid the primary adhesion of living planktonic microbial cells from the surroundings. In general,
this can be achieved by either repelling or killing the approaching cells (see Figure 6). Repelling microbes
was realized with hydrogel coatings mostly based on PEG or similar hydrogel forming polymers, by highly
negatively charged polymers or ultra-hydrophobic modifications. The killing of microbes can be achieved
by either releasing a biocide from a matrix, which is either previously embedded or actively formed for
instance by formation of ROS by photocatalytic TiO2. Alternatively, surfaces can be rendered contact-active
antimicrobial upon tethering antimicrobial polymers [159]. In the following, only surfaces with attached
antimicrobial polymers and multiple working mechanisms will be discussed. Further biocide release systems
in general and microbe repelling surfaces are extensively discussed in recent reviews [160].
References and Notes
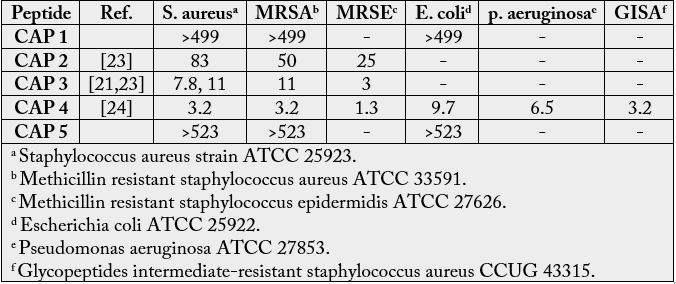
We can conclude from inspecting the table that none of the structures show strand selective eradication.
However, CAP4 seems to be the most efficient one to eradicate both Gram positive and Gram negative
bacteria [161].
Based on these findings, A commercials enterprise which centers on LTX-109 [162] (CAP4 above). LTX 109 was found to be a potent fungicide as well [163]. The company that develops LTX-109, LYNTIX BIOPHARMA [164] is currently focusing on cancer research.
Conclusion
The conclusion of the Authors that small antibacterial agents, surrogates of antimicrobial fragments of
antimicrobial peptide should be developed into novel agent to combat bacterial infections. At first, those
novel agents should serve as antiseptic agent for hygiene purposes and topical infection treatment. In further
view, some of these could serve as a basis for the very urgently needed therapeutic agents for the currently
killing infections: sepsis, post-surgery infections and skin infections and burns. The current situation in
antimicrobial peptides based on natural product from amphibians and reptiles, may yield those agents that
are suitable for antibacterial treatment.
The emergence of enterprises like LYTIX based on agents like LTX-109 in encouraging and spreading hope in the current grim situation
Bibliography

Hi!
We're here to answer your questions!
Send us a message via Whatsapp, and we'll reply the moment we're available!