Biography
Interests
Tayyib, N., Alsolami, F., Asfour, H. I., Ramaiah, P., Ahmed, E. E. & Lindsay, G. M.*
Faculty of Nursing, Umm Al-Qura University, Kingdom of Saudi Arabia
*Correspondence to: Dr. Lindsay, G. M., Faculty of Nursing, Umm Al-Qura University, Kingdom of Saudi Arabia.
Copyright © 2023 Dr. Lindsay, G. M., et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Everyday life has experienced an increase in the availability of mechanisms to measure health data and activities associated with health and well-being. The concept is not new as is evidenced by the historical availability and use of domestic body weight monitoring scales. The invention of Wi-Fi and digital processing of human activity has transformed the population’s capability to assess and monitor a large and growing body of health-related data. The data can be viewed locally, can be interpreted and stored and in some cases for linkage to remote consultation and/or consultation services. The emergence of artificial intelligence (AI), a capability for the analysis and interpretation of meaning in datasets, is central to the functionality of many of the outputs from health device. AI-powered devices are integrated into many of the anticipatory functions available on mobile phones, ‘SMART’ appliances and in searching for subjects via the internet guided by an analysis of your previous use, demographics and perhaps unknown factors identified for the user.
In the main much of the personal health data generated by the general public and patients is not formally used within the healthcare system, but opportunities to use such data as part of an integrated health partnership are gradually gaining traction. It is estimated that trends in the use of a range of existing and in-development medical devices will stimulate the rise in patient generated health data and the potential for costly hospital-based care to be conducted in less costly settings in the community or at home [1].
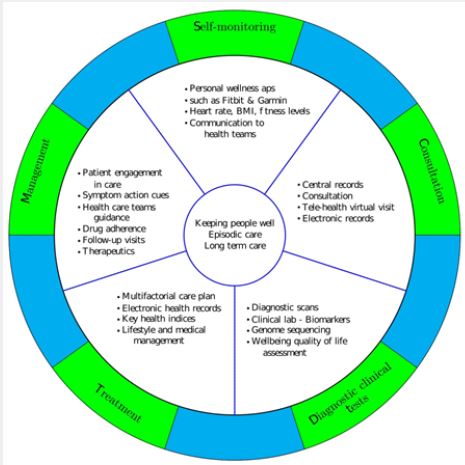
This short report will present, from the public health perspective of ‘keeping people well’, an overview of the range of personal health data that can currently be monitored through accessible personal portable devices, and the emerging assessment and interventional digital devices that are becoming available (Figure 1). The report will also present some of the latest technologies available in the health care setting to assess and intervene to improve health and well-being. What is common to all these processes is that they generate different forms of health data. The potential for greater integration of such diverse data sources is highlighted and the potential challenges for reliability, validity and privacy in centralising data storage.
Self Monitoring
Health monitoring by the individual has expanded greatly in line with technological developments and the
range of digital health biomarkers now accessible. Digital biomarkers can be defined as
‘data that consumers directly collect about health or disease management through digital health technologies to explain, influence and/or predict health-related outcomes’
The most common way to measure ‘biomarkers’ in healthcare is through electronic devices that consumers can wear, the most common of which are Fitbits and smart watches (Wearable technology [2]). Most of these devices are designed to collect a mix of quantitative and qualitative data about their user’s personal health (sleep patterns, stress, mood, feelings of well-being, daily physical activity, diet, body weight, reminders, access to virtual consultations and objective data on cardiac function (heart rate & rhythm, blood pressure); respiratory rate, O2 saturation levels and temperature while the individual is free-living. The monitoring capability can be integrated into wrist watches or specific probes such as self-adhesive patches. Often these devices can also transmit this health information to a doctor or other healthcare professional in real time. The devices offer the ability to store, track, and share personal data with their physician while also providing insights into how personal habits affect the patient’s blood pressure, heart rate and other parameters of health. The aim is to optimize treatments (HeartGuide[3]; Philips’ wearable biosensor [4]; The Move ECG can measure an electrocardiogram, transmit the reading to the user’s physician, and also detect atrial fibrillation.
Consultation
The nature of the consultation has a strong emphasis on a shared care partnership with the patient having
greater involvement in the interchange of information and prioritisation of health issues. The patient can
be equipped with a range of objective and subjective data about their health, thus providing a fundamental
shift in the nature of the healthcare provided by the patient/practitioner consultation. The informed patient
has the capability to engage in a more proactive manner in their contribution to their future health plans.
The electronic health record is used to compile details of the patient’s health data, diagnostics, treatment
plans and has the potential to integrate these patient data into their electronic health record. However, there
remains concern about the validity of patient device data as the context of its collection may be unclear.
Attention to issues of standardisation of the context of data collection would enhance the interpretation
and meaning attributed to device generated data. Under development are new transcribing technologies to
capture the content of the consultation in a format that can be synchronised with electronic patient record
systems which could streamline and improve the efficiency of the consultation by integrating all sources of
health data in a systematic manner into the EHR [6]. Protecting systems for confidentiality of data remains
a matter to be fully resolved.
Diagnostic Laboratory Testing at Home
A selected laboratory profile of blood biochemistry is possible including urea and electrolytes, FBC and Lab
on the skin [7]. This type of wearable device is a patch which is fixed directly onto the skin in an accessible, but comfortable, muscle area of the body such as the upper outer arm. The patch is constructed from microneedles,
one side of which interfaces with the dermal layer of the skin and the other to a case of electronics.
Different enzymes contacting with the tips of the micro-needles react with glucose, alcohol and lactate in
interstitial fluid. These reactions generate small electric currents, which are analyzed by electronic sensors
and then communicate wirelessly to a specifically designed digital application. The results are displayed in
real time on a smart phone. Thus the client can obtain, for example, blood analysis without the need to visit
the doctor’s office or laboratory. However, there are issues of unnecessary testing, increased anxiety over
values obtained without knowing their potential meaning and cost implications.
Imaging
Recent years has seen an increasing use of Artificial Intelligence (AI) in various radiological fields for the
purpose of detecting and diagnosing diseases through technology such as Computerized Tomography (CT)
and Magnetic Resonance (MR) imaging [8]. AI algorithms in radiology detected pneumonia in patients
with better accuracy and recall than the radiologists who participated in the trial. MRI enhanced with AI
capability has been used in oncology to detect abnormalities in cell characteristics related to disease and
to monitor changes over time. Confidence in the precision of AI findings frees up clinician time for direct
patient care and future care planning.
Artificial intelligence based on the interpretation of chest radiographs using neural networks has shown comparable results to radiologists for the identification of lung nodules, calcium deposits in coronary arteries and pneumonia, through the interpretation of chest imaging, high accuracy for liver fibrosis staging, and improved prediction of overall survival in glioblastoma patients from MRI data. Although the results of Convolutional Neural Networks (CNNs) are often comparable to diagnostic radiologists, it is important to note that their detection tasks are narrow in scope, and like other AI methods, they are dependent on large volumes of quality training and validation data. Another concern is related to the AI interpretation of features in image scanning that previously have not been visible to imaging techniques, but are now identified using neural network tools [9].
However, with respect to the wider use of AI and machine learning, there remains uncertainty around the scope of the learning that this technology can safely attain. When an AI-powered device is designed for a particular use, its capability is fixed for that use. However, if the capability of the AI device can continue learning when faced with new patterns or data, then its territory of knowledge in a specialism could exceed that of human experts in that specialism. For example the AI powered device may be able to identify patterns so small that these were not previously identifiable, and make predictions based on this new deepseated data which would otherwise be undetectable by human operators. It leaves many questions about how the future capabilities of AI are best understood and utilised for the greatest benefit [10]. The FDA has approved several “locked” AI algorithms in the U.S. for use in the areas of cardiology, neurology, diabetic retinopathy, intracranial haemorrhage evaluation based on currently available imaging data [11, 12].
Voice Biomarkers
Following the practices of people-isolation during the COVID-19 pandemic, research has focussed on the
potential use of audio clues (e.g. from the voice, breathing patterns or cough of a patient) to diagnose the
presence/severity of certain conditions. There are studies on determining speech biomarkers in Alzheimer’s,
or detecting COVID-19 from voice/cough and breathing patterns or using voice as a biomarker for chronic
obstructive pulmonary disease. Specifically patients with COPD show measureable changes in voice and
breathing that are significantly correlated with changes in the severity of their symptom, with voice ‘signatures’
that detectable even before the symptoms are present [13].
Diagnosis and Treatment
AI empowered interpretation of large datasets can also help with the diagnosis and treatment of diseases
and other conditions to support physician’s decisions and make recommendations based on analyses of
large amounts of relevant healthcare data held in the large databases generated by electronic health records
(EHRs) including symptom data, medical images, doctor reports and more. Such a volume of data is too
much information for humans to process efficiently, to determine what’s worked in the past for similar patient
cohorts. The large scale use of AI applied to fast pattern recognition and the establishment of associations
between input and output variables can help to improve and speed up decision-making and improve
health outcomes. Improvements in diagnostic and treatment processes remain a priority. According to the
Institute of Medicine at the National Academies of Science, Engineering and Medicine [14], “diagnostic
errors contribute to approximately 10 percent of patient deaths” and from 6 to 17 percent of hospital complication.
Some of the latest and most sophisticated digital-powered therapeutics involve robotic systems. Many systems have been developed to work alongside expert clinicians in specific surgical specialisms. Surgical robotics is a subsector of digital healthcare technologies that is experiencing significant growth as robotics becomes the “standard of care” in several areas. Examples include surgery and reconstruction of hips, knees and spines. Key roles involve the precision alignment of surgical sites, targeted interventions and the specific orientation of the patient for specific stages of a procedure. This has proved to be highly successful in brain and spinal surgeries [15] and in tumour removal near vital anatomical structures [16].
Social Robots
Companion robots have been developed to address isolation and loneliness among older vulnerable adults.
These robots are equipped with a video display, camera, microphone, voice active telephone and speaker. The
robots function as social assistants, connecting people with each other. Users can be reminded of appointments
and tasks. Some models provide cognitive stimulation and encourage the user to exercise. Improving
safety is another benefit, as the robot can call for help if the user falls or cries for help. Other functions of
robots can be to provide food and drinks, medications on time and mobility support or transport [17].
The drive and funding invested in the medical devices sector remains robust with evidence of new frontiers. For example, trans-catheter mitral and tricuspid therapies are in the early stages of substantial growth with revenues projected to increase from $1 billion in 2021 to $5 billion by 2028 [18].
Management via Virtual Clinics
The ability for physicians to engage with patients via two-way high-definition (HD) videoconferencing
paves the way for more types of virtual visits than ever before. Patients can now access a new range of treatment
possibilities from their own location by using a smart phone, tablet or PC. Some types of virtual care
that are now available include remote patient monitoring via connected devices, nutrition programs, mental
and behavioural health options and pharmaceutical management. Many organizations build patient portals
that allow patients to communicate with providers by asking questions, requesting medication refills and
scheduling appointments - all virtually. Levels of use have largely stabilized since their peak during the
COVID-19 pandemic and now range from 13 to 17 percent across all specialties. Higher levels are seen in
specialisms of psychiatry and substance abuse with the lowest levels in opthalmics and orthopaedics [19].
These latter specialities are likely to have a higher need for physical examination, particularly in an acute
situation.
Health Data Generation and Utilisation
The processes and some specifics involved in keeping people well (Fig. 1) highlight the sources of data generated
and the creation of a health ‘digital footprint’. The exponential rise in data-generating devices with
the growth of digital enhanced healthcare assessments, diagnostics and interventions has contributed to a
large increase in the amount of health data within the healthcare system. Much becomes available through
AI powered applications which could be used in further problem solving in diagnosis and treatments for
individual patients. Examples of data sources include information from electronic medical records (medical
imaging, patient history, etc.), genetic as well as patient-generated data. According to a white paper by
Cloud Angels, 2850 exabytes (one exabyte = one billion gigabytes) of health data were generated in 2021
[20]. Just to put this into context, the same figure was 153 exabytes in 2013, which means that the volume of
health care data has increased at an annual rate of approximately 44% over the period from 2013 to 2021. In
order to make meaningful use of these data there needs to be ‘joined-up’ systems to capture and store data
in a way that they can be meaningfully retrieved for direct use or for the training of AI powered devices.
Conclusion
To summarize, the current health care landscape is increasingly using digital powered tools for health assessment,
monitoring, diagnostics and treatment. At each point along these pathways data are collected and
stored for future use for the individual’s personal use and also for the greater body of healthcare data that
is stored on health servers. There is growing recognition that to take maximum advantage of this accessible
data requires systems in place for its coordination and management.
As the movement towards ‘healthcare at home’ pushes forward as the population deals with increasing levels of chronic disease that can be managed beyond the acute care setting. Digital health support shows clear potential in realisation of these goals. Such client groups with stable conditions, but sensitive to both medical therapy adherence and lifestyle habits, could benefit from the capabilities offered by relevant wearable health–focused digital devices. In order for such data generated over a wider range of settings and devices to be compatible with and contribute to a synchronised larger dataset, guidelines should be compiled to help provide advice on consistency and context stability when measuring digital biomarkers.
Bibliography

Hi!
We're here to answer your questions!
Send us a message via Whatsapp, and we'll reply the moment we're available!