Biography
Interests
Mohamed Adel ElBasiony1,2, Ramy Hassan Agwa1,3*, Ahmed Abdelaziz4, Reham Soliman2,5 & Gamal Shiha1,2
1Hepatology and Gastroenterology Unit, Department of Internal Medicine, Faculty of Medicine, Mansoura
University, Egypt
2Egyptian Liver Research Institute and Hospital (ELRIAH), Sherbin, El-Mansoura, Egypt
3Department of Internal Medicine, Albaha University, Kingdom of Saudi Arabia
4Department of Gastroenterology and Hepatology , Alazhar University Damietta, Egypt
5Department of Tropical Medicine, Faculty of Medicine, Port Said University, Port Said, Egypt
*Correspondence to: Dr. Ramy Hassan Agwa, Hepatology and Gastroenterology Unit, Department of Internal Medicine, Faculty of Medicine, Mansoura University, Egypt.
Copyright © 2020 Ramy Hassan Agwa, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Brunner’s gland hyperplasia is a rare gastrointestinal (GI) tumor presenting as bleeding or obstructing polyp, usually duodenal. We report a 55 years cirrhotic Egyptian male that presented with repeated attacks of melena, sometimes associated with hematemesis. The bleeding repeated twice to thrice per week. Esophago-gastro-duodenoscope (EGD) showed a duodenal polyp at the second part of the duodenum (D2) and 2 small non-risky esophageal varices. Endoloop was applied at the base of the polyp then removal of the polyp was done using hot snaring. Histopathological examination of the polyp showed that it was Brunner’s gland hyperplasia. Patient followed up for 2 months and the bleeding didn’t recur.
Abbreviations
BGA Brunner’s gland adenoma
CT Computed Tomography
D1 The duodenal bulb
D2 The second part of the duodenum
EGD Esophago-gastro-duodenoscope
GI Gastrointestinal
HCV Hepatitis C virus
Introduction
Primary duodenal tumors are infrequent findings. They account for about 1% of GI tumors [1]. Brunner’s
gland adenoma (BGA) is an extremely rare primary tumor of the duodenum with incidence as low as 0.01%.
It is a benign small bowel neoplasm, also called Brunner’s gland hyperplasia or hamartoma [2]. BGA are
usually found in the duodenal bulb (D1) or D2. They may be discovered as incidental finding during EGD
or radiological examination. BGA clinically present by gastrointestinal bleeding, gastric outlet/ duodenal
obstruction, abdominal pain or intussusception [3,4]. Rarely, BGA may show dysplastic changes (2.1%) or
invasive carcinoma (0.3%) [5,6].
Case Description
A 55 years old patient presented with repeated attacks of upper GI bleeding causing black tarry stools
(melena), sometimes with hematemesis. The bleeding was repeated 2 to 3 times per week over a period of
about 3 months. Clinical assessment showed a clinically stable patient without relevant findings. Laboratory
investigations showed anemia (hemoglobin of 9.5gm. /dl, MCV 65), platelets count 108k/dl, leukocytic
count 7.6k/dl, Hepatitis C virus (HCV) antibody positive, HCV PCR negative, ALT 34u/dl, AST 28u/dl, serum albumin 3.5gm/dl, serum bilirubin 0.9mg/dl, serum creatinine 0.8mg/dl, blood glucose 90mg/dl,
negative HBs antigen and negative HIV antibody. Abdominal ultrasonography showed normal sized liver
with cirrhotic echo-pattern and splenic size of 14cm. No relevant medical or family history found. Patient
received successful anti-HCV treatment 1 year ago with negative HCV PCR on follow up.
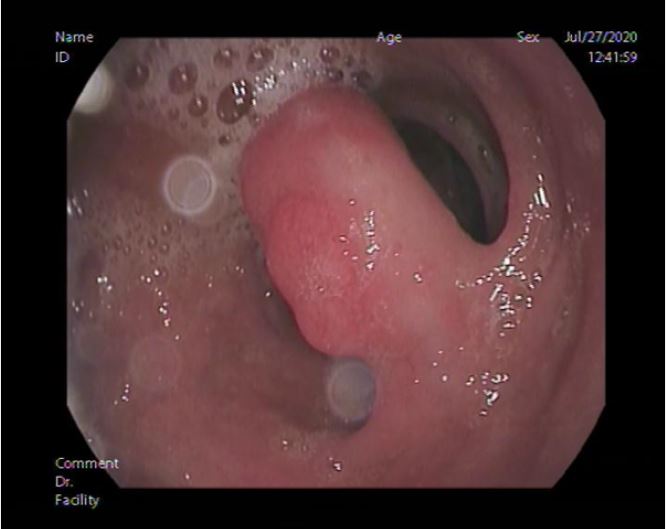
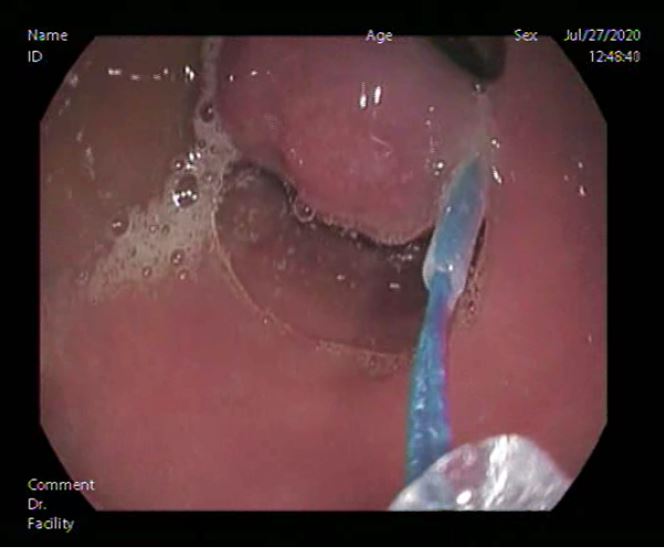
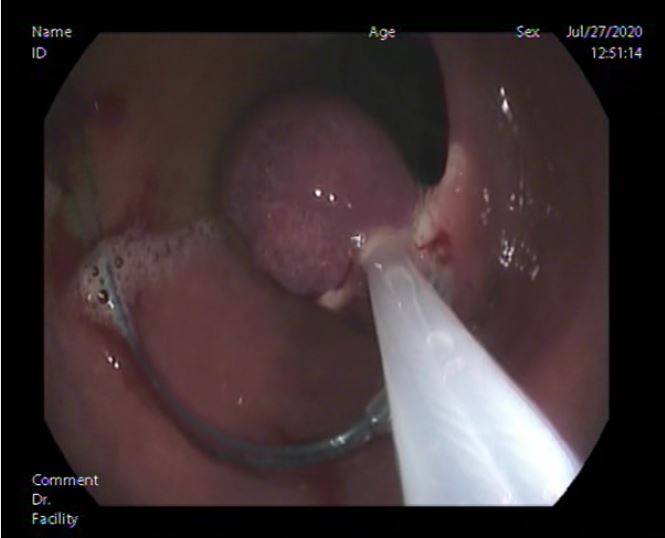
Esophago-gastro-duodenoscope showed 2 small non-risky esophageal varices mild antral gastritis and a polyp about 2x1.5cm size was found at the posterior wall of D2 with inflamed duodenal mucosa (shown in Fig. 1). Endoscopic Endoloop assisted polypectomy was decided. First; Endoloop was applied through the scope to prevent bleeding or perforation (shown in Fig. 2). Snare was secured around the polyp base, just distal to the Endoloop, and hot snaring was used for polypectomy (shown in Fig. 3 & 4). Histopathological examination of the polyp revealed polypoidal tissue structure covered by duodenal mucosa with focal surface erosions and focal regenerative changes. The core of the polyp is formed of lobular proliferation of
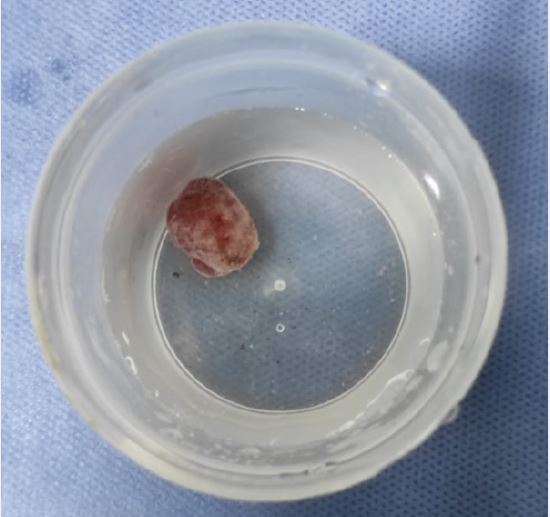
Brunner’s gland. These are lined by mucin secreting cells without atypia. The stroma showed scattered mixed inflammatory cells. No detected cellular atypia or malignancy. This picture was consistent with Brunner’s gland adenoma (shown in Fig. 5,6).
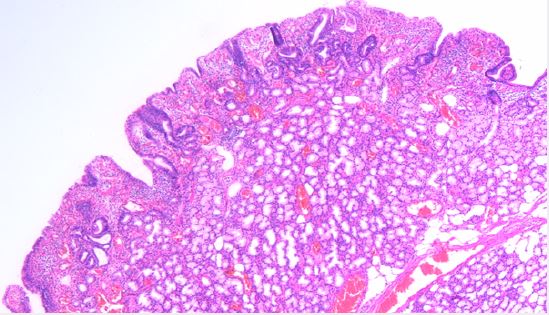
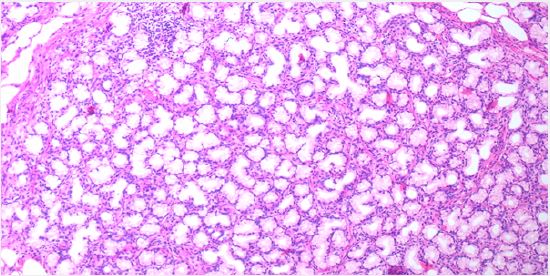
Follow up of the patient was uneventful without complications, namely bleeding or perforation. Upper GI bleeding didn’t recur over a follow up period of about a month.
Discussions
Our case is a 55 years old male with previous chronic HCV infection that was treated before 1 year and the
patient is cirrhotic. The presentation was that of upper GI bleeding by melena. EGD showed 2 small nonrisky
esophageal varices with no signs of bleeding from them and also revealed D2 polyp. The main differential
diagnoses are gastrointestinal stromal tumor, lymphoma, carcinoid, Peutz-Jeghers polyps, prolapsed pyloric
mucosa, or aberrant pancreatic tissue. Endoloop assisted endoscopic removal of the polyp was done using
hot snare. Traditional endoscopy of pinching biopsies is usually negative since the biopsy forceps are unable
to reach the tumoral tissue localized completely in the submucosa layer [7]. Histopathological examination
of the retrieved polyp revealed lobular proliferation of the mucin secreting Brunner’s gland at the core of the
polyp making the diagnosis of BGA.
Brunner glands are branched acinotubular glands in the submucosa of the duodenum. They are located mainly in the duodenal bulb, proximal duodenum, and, with a progressive decrease in number and size, in the distal duodenum. They secrete an alkaline mucin that coats the duodenal epithelium, acting as a protector from the acid chyme of the stomach [8]. Brunner’s gland adenoma is a rare tumor-like lesion, mostly present in middle age [3]. It was first described by Curveilhier, 1835 [9]. Regarding the size, they are described as Brunner’s gland hyperplasia when the size is < 1cm [10]. The majority of cases are polypoidal lesions 1-2cm in size named BGA with few lesions reaching up to 5 cm and are called giant BGA [11-14]. Dysplastic changes were seen in 2.1% and invasive carcinoma in 0.3% of all Brunner’s gland hyperplasia [6].
The pathogenesis of BGA is still unclear. It is thought that increased acid secretion may stimulate Brunner’s gland growth and hyperplasia [15]. A second hypothesis suggests that this lesion is of inflammatory origin due to the presence of a dense inflammatory cell infiltration [16]. Helicobacter pylori infection may be accused of causing BGA, but the relationship had not been proven due to the rarity of BGA and the widespread of helicobacter pylori infection [17,18].
Cases with BGA may be asymptomatic or may have non-specific symptoms and the lesions are discovered accidentally during radiological examinations or EGD. Symptomatic patients may show GI bleeding or obstruction symptoms. The bleeding may be chronic leading to iron deficiency anemia or bouts of melena with or without hematemesis [3,19]. Other presenting features may be that of pancreatitis, biliary obstruction, mimicking pancreatic cancer, annular duodenal stricture or gastric outlet obstruction [20-22].
Diagnosis of BGA is usually not easy and it is an incidental finding in many situations. Radiological examinations like ultrasonography and Computed tomography (CT) can detect large lesions [14]. Another value of CT is to confirm the absence of extra-luminal extension of BGA [23]. Upper GI Barium studies show BGA as smooth-walled polypoid filling defects usually in the duodenal bulb [24]. Radiological findings are not specific. The differential diagnosis includes leiomyoma, lipoma or lymphoma and another investigatory tool is needed [23]. EGD can show the lesion’s location and features but superficial biopsies are not diagnostic. So; deep, excision or surgical biopsies are often needed [25,26]. In our case the diagnosis was made by endoscopic excision biopsy that showed the stroma of the polyp with Brunner’s gland. Endoscopic ultrasound (EUS) can show the origin, extent and vascularity of the lesion with the ability of deep biopsies [27]. Endoscopic or surgical excision of the lesion is usually the treatment required in most symptomatic cases. Endoscopic polypectomy represents the ideal approach, which is more cost-effective and less invasive of the abdominal surgery [28,29]. In our case; Endoloop assisted excision of the lesion using hot snare was done without any complications to consider and short time procedure with rapid recovery. Reports show that there is no evidence of recurrence after endoscopic or surgical removal and the prognosis is generally good [25,30].
Conclusions
In conclusion; BGA is a rare benign tumor, mostly arising from the duodenum. The most common clinical
presentation is bleeding or obstruction in symptomatic cases. Endoloop assisted endoscopic removal of the
lesion is a safe technique with good outcome. BGA can be the source of upper GI bleeding even in cases
with small esophageal varices.
Acknowledgements
Thanks, and greetings are granted to all the Egyptian people who donated to support ELRIAH to do its
work for supporting Egyptian liver.
Conflicts of Interests
The authors have no conflicts of interest to declare.
Funding Sources
This work was supported by Egyptian liver research institute and hospital (ELRIAH) that was founded
by Association of liver patient care which is non-governmental non-profit organization charity since 1997
which depends on donations from ordinary people.
Bibliography

Hi!
We're here to answer your questions!
Send us a message via Whatsapp, and we'll reply the moment we're available!